If a given mass of gas occupies a volume of $10\, cc$ at $1$ atmospheric pressure and temperature of $100°C(373.15\, K).$ What will be its volume at 4 atmospheric pressure; the temperature being the same .... $cc$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$) - 2To raise the temperature of a certain mass of gas by $50^{\circ} C$ at a constant pressure, $160$ calories of heat is required. When the same mass of gas is cooled by $100^{\circ} C$ at constant volume, $240$ calories of heat is released. How many degrees of freedom does each molecule of this gas have (assume gas to be ideal) $?$View Solution
- 3View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will
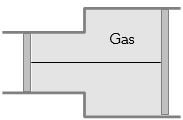
- 4A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 5We write the relation for Boyle's law in the form $PV = C$ when the temperature remains constant. In this relation, the magnitude of $C$ depends uponView Solution
- 6View SolutionThe absolute zero is the temperature at which
- 7An ideal gas is enclosed in a container of volume $V$ at a pressure $P$. It is being pumped out of the container by using a pump with stroke volume $v$. What is final pressure in container after $n$-stroke of the pump? (assume temperature remains same)View Solution
- 8A closed vessel contains $10 g$ of an ideal gas $X$ at $300 \ K$, which exerts $2$ atm pressure. At the same temperature, $80 g$ of another ideal gas $Y$ is added to it and the pressure becomes $6 atm$. The ratio of root mean square velocities of $X$ and $Y$ at $300 \ K$ isView Solution
- 9A gaseous mixture contains equal number of hydrogen and nitrogen molecules. Specific heat measurements on this mixture at temperatures below $100\, K$ would indicate that the value of $\gamma $ (ratio of specific heats) for this mixture isView Solution
- 10The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
