A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume occupied by the molecules contained in $4.5\,kg$ water at $STP$, if the intermolecular forces vanish away is ........ $m ^{3}$View Solution
- 2Under which of the following conditions is the law $PV = RT$ obeyed most closely by a real gasView Solution
- 3If $\alpha$ moles of a monoatomic gas are mixed with $\beta$ moles of a polyatomic gas and mixture behaves like diatomic gas, then [neglect the vibrational mode of freedom]View Solution
- 4Initially a gas of diatomic molecules is contained in a cylinder of volume $V _{1}$ at a pressure $P_{1}$ and temperature $250\, K$. Assuming that $25 \%$ of the molecules get dissociated causing a change in number of moles. The pressure of the resulting gas at temperature $2000\, K ,$ when contained in a volume $2 V _{1}$ is given by $P _{2}$. The ratio $\frac{P _{2}}{ P _{1}}$ is.View Solution
- 5$N$ molecules each of mass $m$ of gas $A$ and $2N$ molecules each of mass $2m$ of gas $B$ are contained in the same vessel at temperature $T.$ The mean square of the velocity of molecules of gas $B$ is ${v^2}$ and the mean square of $x$ component of the velocity of molecules of gas $A$ is ${w^2}$. The ratio $\frac{{{w^2}}}{{{v^2}}}$ isView Solution
- 6An ideal gas with heat capacity at constant volume $C_V$ undergoes a quasistatic process described by $p V^{\alpha}$ in a $p-V$ diagram, where $\alpha$ is a constant. The heat capacity of the gas during this process is given byView Solution
- 7Two bulbs of identical volumes connected by a small capillary are initially filled with an ideal gas at temperature $T$. Bulb $2$ is heated to maintain a temperature $2 T$, while bulb $1$ remains at temperature $T$. Assume throughout that the heat conduction by the capillary is negligible. Then, the ratio of final mass of the gas in bulb $2$ to the initial mass of the gas in the same bulb is close toView Solution
- 8An insulated container contains $4\, moles$ of an ideal diatomic gas at temperature $T$. Heat $Q$ is supplied to this gas, due to which $2\, moles$ of gas are dissociated into atoms but temperature of the gas remains constant. Then:View Solution
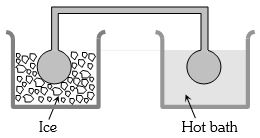
- 9Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$View Solution
- 10Under an adiabatic process, the volume of an ideal gas gets doubled. Consequently the mean collision time between the gas molecule changes from $\tau_{1}$ to $\tau_{2} .$ If $\frac{C_{p}}{C_{v}}=\gamma$ for this gas then a good estimate for $\frac{\tau_{2}}{\tau_{1}}$ is given by :View Solution
