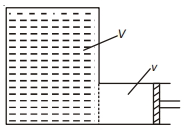
An ideal gas is enclosed in a container of volume $V$ at a pressure $P$. It is being pumped out of the container by using a pump with stroke volume $v$. What is final pressure in container after $n$-stroke of the pump? (assume temperature remains same)
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Air is filled in a bottle at atmospheric pressure and it is corked at $35°C.$ If the cork can come out at $3$ atmospheric pressure than upto what temperature should the bottle be heated in order to remove the cork ...... $^oC$View Solution
- 2The mean free path of gas molecules depends on $(d =$ molecular diameter$)$View Solution
- 3An insulated container contains $4\, moles$ of an ideal diatomic gas at temperature $T$. Heat $Q$ is supplied to this gas, due to which $2\, moles$ of gas are dissociated into atoms but temperature of the gas remains constant. Then:View Solution
- 4Consider a mixture of $n$ moles of helium gas and $2 n$ moles of oxygen gas (molecules taken to be rigid) as an ideal gas. Its $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}$ value will beView Solution
- 5A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 6Two gases-argon (atomic radius $0.07 \;\mathrm{nm}$,atomic weight $40$ ) and xenon (atomic radius $0.1\; \mathrm{nm},$ atomic weight $140$ ) have the same number density and are at the same temperature. The raito of their respective mean free times is closest toView Solution
- 7View SolutionTwo moles a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is:
- 8$\frac{1}{2} $ mole of helium gas is contained in a container at $S.T.P.$ The heat energy needed to double the pressure of the gas, keeping the volume constant (specific heat of the gas $ = 3\,J\,g{m^{ - 1}}\,{K^{ - 1}})$ is ...... $J$View Solution
- 9View SolutionWhich of the following statements about kinetic theory of gases is wrong
- 10$70$ calories of heat are required to raise the temperature of $2$ moles of an ideal gas at constant pressure from $30°C$ to $35°C.$ The amount of heat required to raise the temperature of same gas through the same range $(30°C$ to $35°C)$ at constant volume ..... $cal$ $(R = 2 \,cal/mol/K)$View Solution