If in the experiment of Wheatstone's bridge, the positions of cells and galvanometer are interchanged, then balance points will
AIIMS 2017, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
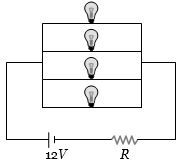
- 1Four identical electrical lamps are labelled $1.5\,V$, $0.5\,A$ which describes the condition necessary for them to operate at normal brightness. A $12\,V$ battery of negligible internal resistance is connected to lamps as shown, thenView Solution
- 2Infinite number of cells having $emf$ and internal resistance $\left( {E,r} \right)$, $\left( {\frac{E}{n},\frac{r}{n}} \right)$, $\left( {\frac{E}{{{n^2}}},\frac{r}{{{n^2}}}} \right)$, $\left( {\frac{E}{{{n^3}}},\frac{r}{{{n^3}}}} \right)$..... are connected in series in same manner across an external resistance of $\frac{{nr}}{{n + 1}}$ . Current flowing through the external resistor isView Solution
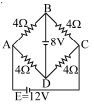
- 3The given figure shows $a$ network of resistances and $a$ battery. Which of the two batteries is getting charged?View Solution
- 4View SolutionWhen cells are connected in parallel, then
- 5$A$ potentiometer wire has length $10\, m$ and resistance $10\,\Omega$ . It is connected to a battery of $EMF$ $11\, volt$ and internal resistance $1\, \Omega$ , then the potential gradient in the wire is ............... $V/m$View Solution
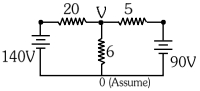
- 6The value of current in the $6 \,\Omega$ resistance is $....\,A$View Solution
- 7It is preferable to measure the $e.m.f.$ of a cell by potentiometer than by a voltmeter because of the following possible reasons.View Solution
$(i)$ In case of potentiometer, no current flows through the cell.
$(ii)$ The length of the potentiometer allows greater precision.
$(iii)$ Measurement by the potentiometer is quicker.
$(iv)$ The sensitivity of the galvanometer, when using a potentiometer is not relevant.
Which of these reasons are correct? - 8A cell of $emf\;4\,V$ and internal resistance $0.5\,\Omega$ is connected to a $7.5\,\Omega$ external resistance. The terminal potential difference of the cell is $.....\,V$.View Solution
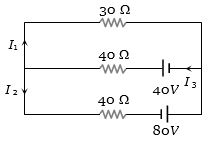
- 9In the given circuit the current $I_1$ is .............. $A$View Solution
- 10The internal resistance of a primary cell is $4\, ohm$. It generates a current of $0.2\, amp$ in an external resistance of $21\, ohm$. The rate at which chemical energy is consumed in providing the current is .............. $J/s$View Solution
