If radius of $O _{2}$ molecule $=40 \mathring A, T =27^{\circ} C$ and $P =1 \,atm .$ Find the time of relaxation.
AIIMS 2019, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Vessel $A$ is filled with hydrogen while vessel $B,$ whose volume is twice that of $A,$ is filled with the same mass of oxygen at the same temperature. The ratio of the mean kinetic energies of hydrogen and oxygen isView Solution
- 2View SolutionSpecific heats of monoatomic and diatomic gas is same than which relation is valid
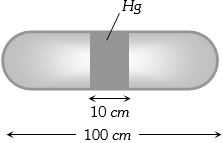
- 3A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 4A jar has a mixture of hydrogen and oxygen gas in the ratio of $1 : 5$. The ratio of mean kinetic energies of hydrogen and oxygen molecules isView Solution
- 5A mixture of one mole of monoatomic gas and one mole of a diatomic gas (rigid) are kept at room temperature $\left(27^{\circ} \mathrm{C}\right)$. The ratio of specific heat of gases at constant volume respectively is:View Solution
- 6View SolutionFor ideal gas, which statement is not true
- 7A container contains $32 \,g$ of $O _2$ at a temperature $T$. The pressure of the gas is $P$. An identical container containing $4 g$ of $H _2$ at a temperature $2 T$ has a pressure of ............View Solution
- 8The mean free path for a gas, with molecular diameter $d$ and number density $n$ can be expressed as :View Solution
- 9In the absence of intermolecular force of attraction, the observed pressure $P$ will beView Solution
- 10Air is filled in a bottle at atmospheric pressure and it is corked at $35°C.$ If the cork can come out at $3$ atmospheric pressure than upto what temperature should the bottle be heated in order to remove the cork ...... $^oC$View Solution
