ઈલેકટ્રોનુરાગી $\mathop N\limits^ \oplus {O_2}$ નીચેનામાંથી કોની સાથે પ્રક્રિયા કરે છે. $\mathop N\limits^ \oplus {O_2}$એ કયા કિસ્સામાં મેટા સ્થાનમાં પ્રક્રિયા કરશે.
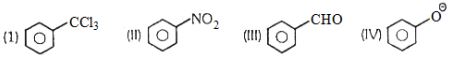
Diffcult
b
$- CCl_3$ , $- NO_2$ & - $CHO $ સમુહોના કારણે $ - m $ અસરના ધરાવે છે જે મેટા નિદેશક છે.
$- CCl_3$ , $- NO_2$ & - $CHO $ સમુહોના કારણે $ - m $ અસરના ધરાવે છે જે મેટા નિદેશક છે.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1બેઇઝ પ્રબળતાના ઘટતા ક્રમમાં નીચેના ક્રમ આપોView Solution
$(A)\, CH_3 - CH_2 - C \equiv C^-$ $(B) \,CH_3 -CH_2 - S^-$
$(C) \,CH_3 - CH_2 - CO^-_2$ $(D)\, CH_3 -CH_2 - O^-$ - 2View Solutionનીચેનામાંથી મુકતમુલક નો સ્થાયિતાનો વધતો ક્રમ કયો છે?
- 3View Solutionનીચેના પદાર્થો માટે એસિડિક ક્ષમતાનો ક્રમ કયો છે ?
- 4View Solutionનીચેનામાંથી કયા સિગ્મા બંધ અતિસન્યુગમન માં ભાગ લે છે
- 5View Solutionનીચેનામાંથી કોણ મહત્તમ કેન્દ્રાનુરાગીતા ધરાવે છે ?
- 6View Solutionનીચે આપેલા પૈકી સૌથી વધુ સ્થિર ક્રાર્બોશાશાયન (કાર્બોકેટાયન) શોધો.
- 7View Solutionકાર્બોકેશાયન (કાર્બોકેટાયન) નો આકાર. . . . . . છે.
- 8ક્લોરોબેન્ઝિન માધ્યમમાં જુદા જુદા એમાઈનનો સાચો બેઝિકતા ક્રમ કયો છે ?View Solution
$(A) \,\,CH_3-CH_2-NH_2$
- 9View Solutionસૂર્યપ્રકાશ વડે કેવા પ્રકારનું વિભાજન શક્ય છે ?
- 10નીચેના માટે ઈલેકટ્રોન અનુરાગી માટે ઘટતો પ્રક્રિયાશીલતા ક્રમ કયો છે ?View Solution
$(1)$ બેન્ઝીન $(2)$ ટોલ્યુઈન $(3)$ ક્લોરો બેન્ઝિન $(4)$ ફિનોલ
