In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$
Easy
(c) $\Delta Q = \Delta U + \Delta W = ({U_f} - {U_i}) + \Delta W$
==> $30 = ({U_f} - 40) + 10$==> ${U_f} = 60J$
==> $30 = ({U_f} - 40) + 10$==> ${U_f} = 60J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 2View SolutionMonoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.
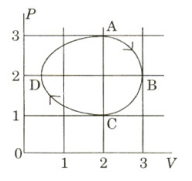
- 3The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA$. The part $ABC$ is a semi-circle and $CDA$ is half of an ellipse. Then,View Solution
$(A)$ the process during the path $\mathrm{A} \rightarrow \mathrm{B}$ is isothermal
$(B)$ heat flows out of the gas during the path $\mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D}$
$(C)$ work done during the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C}$ is zero
$(D)$ positive work is done by the gas in the cycle $ABCDA$
- 4Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 5View SolutionIn a reversible isochoric change
- 6A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
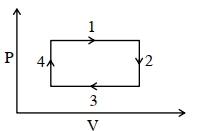
- 7An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
- 8A perfect gas goes from state $A$ to another state $B$ by absorbing $8 \times {10^5}J$ of heat and doing $6.5 \times {10^5}J$ of external work. It is now transferred between the same two states in another process in which it absorbs ${10^5}J$ of heat. Then in the second processView Solution
- 9View SolutionDuring an isothermal expansion of an ideal gas
- 10View SolutionWork done on or by a gas, in general depends upon the
