Monoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.
Medium
For an adiabatic process,
$P V^{\gamma}=$ constant
or
$\frac{P_{1}}{P_{2}}=\left(\frac{V_{2}}{V_{1}}\right)^{\gamma}$
or
$P_{2}=2^{\gamma} P_{1}$
Now, for a monoatomic gas, the value of $\gamma$ is the highest
Thus, for the same change in volume, the monoatomic gas will have the maximum pressure.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monoatomic gas $\left( {\gamma = \frac{5}{3}} \right)$ is suddenly compressed to $\frac{1}{8}$ of its original volume, then the pressure of gas will change to how many times the initial pressure?View Solution
- 2The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$View Solution
- 3$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 4View SolutionIf a gas is heated at constant pressure, its isothermal compressibility
- 5An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
- 6In a carnot engine, the temperature of reservoir is $527^{\circ} C$ and that of $\operatorname{sink}$ is $200 \; K$. If the workdone by the engine when it transfers heat from reservoir to sink is $12000 \; kJ$, the quantity of heat absorbed by the engine from reservoir is $\times 10^{6} \; J$View Solution
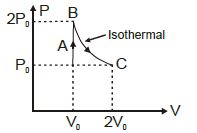
- 7A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
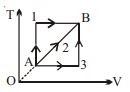
- 8A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution
- 9During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
- 10View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
