Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
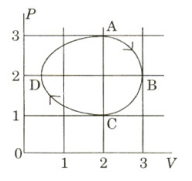
- 2The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA$. The part $ABC$ is a semi-circle and $CDA$ is half of an ellipse. Then,View Solution
$(A)$ the process during the path $\mathrm{A} \rightarrow \mathrm{B}$ is isothermal
$(B)$ heat flows out of the gas during the path $\mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D}$
$(C)$ work done during the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C}$ is zero
$(D)$ positive work is done by the gas in the cycle $ABCDA$
- 3In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
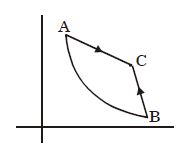
- 4Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
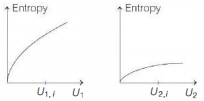
- 5Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
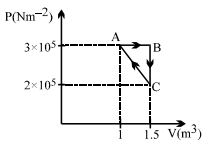
- 6Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
- 7The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
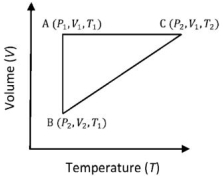
- 8A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 9View SolutionWhen heat in given to a gas in an isobaric process, then
- 10View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
