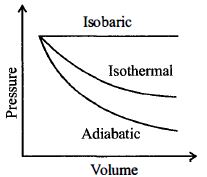
An ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
KVPY 2009, Medium
(c)
Heat supplied at constant pressure is $\Delta Q=n C_p \Delta T$.
Work done by the gas,
$\Delta W=p \Delta V=n R \Delta T$
Ratio of work done to heat supplied is
$\frac{\Delta W}{\Delta Q}=\frac{n R \Delta T}{n C_p \Delta T}$
$=\frac{R}{C_p}=\frac{C_p-C_V}{C_p}$
$=1-\frac{1}{\gamma}$
$=1-\frac{1}{(7 / 5)}=1-\frac{5}{7}=\frac{2}{7}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The latent heat of vaporization of water is $2240 \,J/gm$. If the work done in the process of vaporization of $1\, gm$ is $168\, J$, then increase in internal energy is .... $J$View Solution
- 2During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 3The temperature inside a refrigerator is $t_2 \,^o C$ and the room temperature is $t_1\,^o C.$ The amount of heat delivered to the room for each joule of electrical energy consumed ideally will beView Solution
- 4The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
- 5An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
- 6$n-$ $moles$ of an ideal gas with constant volume heat capacity $C_v$ undergo an isobaric expansion by certain volume. The ratio of the work done in the process, to the heat supplied isView Solution
- 7$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 8A vertical cylinder with heat-conducting walls is closed at the bottom and is fitted with a smooth light piston. It contains one mole of an ideal gas. The temperature of the gas is always equal to the surrounding’s temperature, $T_0$. The piston is moved up slowly to increase the volume of the gas to $\eta$ times. Which of the following is incorrect?View Solution
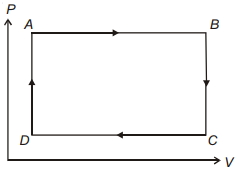
- 9Work done in the given $P-V$ diagram in the cyclic process isView Solution
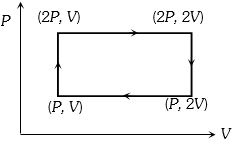
- 10A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution