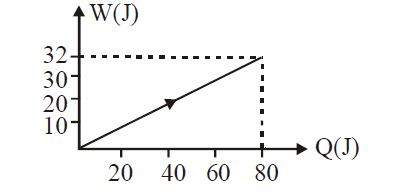
In the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be

Medium
$\frac{W}{Q}=\frac{n R \Delta T}{n C_{p} \Delta T}=\frac{3 2}{80}=\frac{2}{5}$
$5 \mathrm{R}=2 \mathrm{C}_{\mathrm{p}}=2\left[\mathrm{C}_{\mathrm{v}}+\mathrm{R}\right]$
$C_{v}=\frac{3 R}{2}$
$\therefore(1)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn a reversible isochoric change
- 2A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution
- 3Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 4In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 5For $P-V$ diagram of a thermodynamic cycle as shown in figure, process $B C$ and $D A$ are isothermal. Which of the corresponding graphs is correct?View Solution
- 6The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
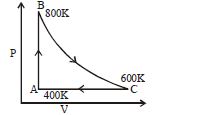
- 7One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 8View SolutionIn the cyclic process shown in the figure, the work done by the gas in one cycle is
- 9A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 10View SolutionWhich of the following statements is correct for any thermodynamic system
