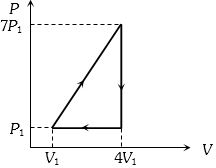
In the cyclic process shown in the figure, the work done by the gas in one cycle is

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
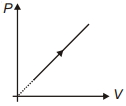
- 2View SolutionDuring the thermodynamic process shown in figure for an ideal gas
- 3During the adiabatic expansion of $2$ moles of a gas, the internal energy of the gas is found to decrease by $2$ joules, the work done during the process on the gas will be equal to ....... $J$View Solution
- 4View SolutionFor a reversible process, necessary condition is
- 5If heat is added at constant volume, $6300\,\,J$ of heat are required to raise the temperature of an ideal gas by $150\,\,K$. If instead, heat is added at constant pressure, $8800$ joules are required for the same temperature change. When the temperature of the gas changes by $300\,\,K$, the internal energy of the gas changes by ..... $J$View Solution
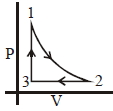
- 6Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 7A thermally insulated container is divided into two parts by a screen. In one part the pressure and temperature are $P$ and $T$ for an ideal gas filled. In the second part it is vacuum. If now a small hole is created in the screen, then the temperature of the gas willView Solution
- 8A diatomic ideal gas is compressed adiabatically to $\frac{1}{32}$ of its initial volume. If the initial temperature of the gas is $T_1$ (in Kelvin) and the final temperature is $a T_1$, the value of $a$ isView Solution
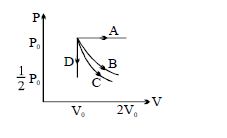
- 9The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process$(es)$ does the temperature of the gas decrease?View Solution
- 10Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

