In the $P-V$ diagram, $I$ is the initial state and $F$ is the final state. The gas goes from $I$ to $F$ by $(i)\,IAF,\,\,(ii)\,IBF,\,\,(iii) ICF$. The heat absorbed by the gas is
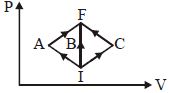

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe process in which no heat enters or leaves the system is termed as
- 2One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal toView Solution
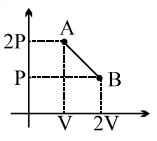
- 3A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
- 4View SolutionA measure of the degree of disorder of a system is known as
- 5The coefficient of performance of a refrigerator is $5.$ If the temperature inside freezer is $-20^o C,$ the temperature of the surroundings to which it rejects heat is ........ $^oC$View Solution
- 6View SolutionFor free expansion of the gas which of the following is true
- 7In an Isobaric process, the work done by a di$-$atomic gas is $10\, J ,$ the heat given to the gas will be (in $J$)View Solution
- 8$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 9When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
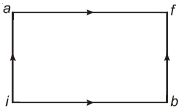
- 10View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
