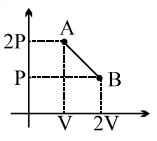
One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal to

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
- 2In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
- 3A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution
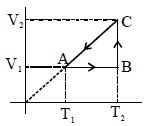
- 4View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
- 5One mole of a gas obeying the equation of state $P(V-b)=R T$ is made to expand from a state with coordinates $\left(P_{1}, V_{1}\right)$ to a state with $\left(P_{2}, V_{2}\right)$ along a process that is depicted by a straight line on a $P-V$ diagram. Then, the work done is given byView Solution
- 6An ideal gas is made to undergo the cyclic process shown in the figure below. Let $\Delta W$ depict the work done, $\Delta U$ be the change in internal energy of the gas and $Q$ be the heat added to the gas. Sign of each of these three quantities for the whole cycle will be (0 refers to no change)View Solution
- 7A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 8A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 9View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 10A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
