$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$
AIIMS 1997, Easy
$\Delta \mathrm{Q}=\Delta \mathrm{U}+\mathrm{P} \Delta \mathrm{V} \quad[\mathrm{PV}=\mathrm{nRT}$
$\Delta \mathrm{Q}=\Delta \mathrm{U}+\mathrm{nR} \Delta \mathrm{T} \quad \mathrm{P} \Delta \mathrm{V}=\mathrm{nR} \Delta \mathrm{T}$
For isothermal change $\Delta \mathrm{T}=0$
$\therefore \Delta Q=\Delta U$
In other words whole of heat supplied is converted into internal energy.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$View Solution
- 2View SolutionIn adiabatic expansion of a gas
- 3A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 4A gas at initial temperature $T$ undergoes sudden expansion from volume $V$ to $2 \,V$. Then,View Solution
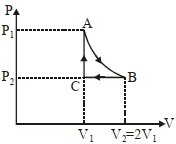
- 5$n$ mole a perfect gas undergoes a cyclic process $ABCA$ (see figure) consisting of the following processes.View Solution
$A \rightarrow B :$ Isothermal expansion at temperature $T$ so that the volume is doubled from $V _{1}$ to $V _{2}=2 V _{1}$ and pressure changes from $P _{1}$ to $P _{2}$
$B \rightarrow C :$ Isobaric compression at pressure $P _{2}$ to initial volume $V _{1}$
$C \rightarrow A$ : Isochoric change leading to change of pressure from $P _{2}$ to $P _{1}$
Total workdone in the complete cycle $ABCA$ is
- 6One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume isView Solution
- 7View SolutionIn an isobaric expansion of an ideal gas, which of the following is zero?
- 8View SolutionWhich statement is incorrect?
- 9Two ideal Carnot engines operate in cascade (all heat given up by one engine is used by the other engine to produce work) between temperatures, $\mathrm{T}_{1}$ and $\mathrm{T}_{2} .$ The temperature of the hot reservoir of the first engine is $\mathrm{T}_{1}$ and the temperature of the cold reservoir of the second engine is $\mathrm{T}_{2} . T$ is temperature of the sink of first engine which is also the source for the second engine. How is $T$ related to $\mathrm{T}_{1}$ and $\mathrm{T}_{2}$, if both the engines perform equal amount of work?View Solution
- 10View SolutionA gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then
