Match List$-I$ with List$-II$
| List$-I$ | List$-II$ |
| $(a)$ Isothermal | $(i)$ Pressure constant |
| $(b)$ Isochoric | $(ii)$ Temperature constant |
| $(c)$ Adiabatic | $(iii)$ Volume constant |
| $(d)$ Isobaric | $(iv)$ Heat content is constant |
Choose the correct answer from the options given below
JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 2$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
- 3View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
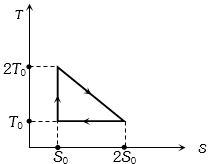
- 4The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
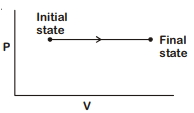
- 5View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
- 6View SolutionThe ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
- 7Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\epsilon_1$ and $\epsilon_2$, respectively. The net efficiency of the combination is given by :View Solution
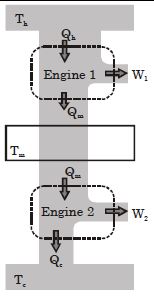
- 8Carnot heat engine works with an ideal diatomic gas and an adiabatic volume expansion ratio $32$ . Then its efficiency is ....... $\%$View Solution
- 9An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 10During an adiabatic compression, $830\, J$ of work is done on $2\, moles$ of a diatomic ideal gas to reduce its volume by $50\%$. The change in its temperahture is nearly..... $K$ $(R\, = 8.3\, J\,K^{-1}\, mol^{-1} )$View Solution
