A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)
JEE MAIN 2021, Diffcult
${T}_{2}=$ sink temperature
$\eta=1-\frac{{T}_{2}}{{T}_{1}}$
$\frac{1}{4}=1-\frac{{T}_{2}}{{T}_{1}}$
$\frac{{T}_{2}}{{T}_{1}}=\frac{3}{4} \ldots$ $(i)$
$\frac{1}{2}=1-\frac{{T}_{2}-58}{{T}_{1}}$
$\frac{{T}_{2}}{{T}_{1}}-\frac{58}{{T}_{1}}=\frac{1}{2}$
$\frac{3}{4}=\frac{58}{{T}_{1}}+\frac{1}{2}$
$\frac{1}{4}=\frac{58}{{T}_{1}} \Rightarrow {T}_{1}=232$
${T}_{2}=\frac{3}{4} \times 232$
${T}_{2}=174\, {K}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An air bubble of volume $v _0$ is released by a fish at a depth $h$ in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density) of water is $\rho$View Solution
- 2At $N.T.P.$ one mole of diatomic gas is compressed adiabatically to half of its volume $\gamma = 1.41$. The work done on gas will be ....... $J$View Solution
- 3The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
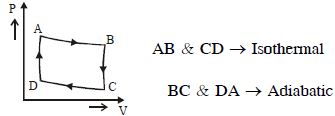
- 4View SolutionWork done per mol in an isothermal change is
- 5View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 6A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 7Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$View Solution

- 8View SolutionThe first operation involved in a Carnot cycle is
- 9Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
- 10Two samples $A$ and $B$ of a gas initially at the same pressure and temperature are compressed from volume $ V$ to $ V/2$ ($A$ isothermally and adiabatically). The final pressure of $ A$ isView Solution
