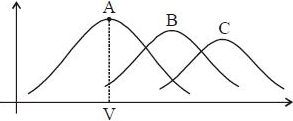
Maxwell distribution function is shown in figure for different gases, which of the following is correct matching?
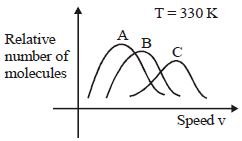

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At ..... $K$ temperature average speed of $O_2$ will be ${\left( {\frac{7}{4}} \right)^{th}}$ of rms speed of $N_2$ at $47\,^oC$View Solution
- 2One kg of a diatomic gas is at a pressure of $8 × 10^4\ N/m^2$. The density of the gas is $4\ kg/m^3$. What is the energy of the gas due to its thermal motion ?View Solution
- 3View SolutionAt constant volume, for different diatomic gases the molar specific heat is
- 4The density of a gas at normal pressure and $27°C$ temperature is $24.$ Keeping the pressure constant, the density at $127°C$ will beView Solution
- 5If temperature of gas increases from $27°C$ to $927°C$ the $K.E.$ will beView Solution
- 6The $r.m.s.$ speed of a certain gas is v at $400\,K.$ The temperature at which the $r.m.s.$ speed becomes two times, will be ...... $K$View Solution
- 7View SolutionA monoatomic gas molecule has
- 8When unit mass of water boils to become steam at $100\,^0C$, it absorbs $Q$ amount of heat. The densities of water and steam at $100\,^0C$ are $\rho_1$ and $\rho_2$ respectively and the atmospheric pressure is $p_0$. The increase in internal energy of the water isView Solution
- 9View SolutionAt a given temperature the root mean square velocities of oxygen and hydrogen molecules are in the ratio
- 10Air is filled in a bottle at atmospheric pressure and it is corked at $35°C.$ If the cork can come out at $3$ atmospheric pressure than upto what temperature should the bottle be heated in order to remove the cork ...... $^oC$View Solution