When unit mass of water boils to become steam at $100\,^0C$, it absorbs $Q$ amount of heat. The densities of water and steam at $100\,^0C$ are $\rho_1$ and $\rho_2$ respectively and the atmospheric pressure is $p_0$. The increase in internal energy of the water is
Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
- 2Two gases occupy two containers $A$ and $B$ the gas in $A$, of volume $0.10\,m ^3$, exerts a pressure of $1.40\,MPa$ and that in $B$ of volume $0.15 m ^3$ exerts a pressure $0.7\,MPa$. The two containers are united by a tube of negligible volume and the gases are allowed to intermingle. Then if the temperature remains constant, the final pressure in the container will be (in MPa)View Solution
- 3An ideal gas filled in a cylinder occupies volume $V$. The gas is compressed isothermally to the volume $V/3$. Now, the cylinder valve is opened and the gas is allowed to leak keeping temperature same. What percentage of the number of molecules should escape to bring the pressure in the cylinder back to its original value?View Solution
- 4If the collision frequency of hydrogen molecules in a closed chamber at $27^{\circ} \mathrm{C}$ is $\mathrm{Z}$, then the collision frequency of the same system at $127^{\circ} \mathrm{C}$ is :View Solution
- 5View SolutionSupposing the distance between the atoms of a diatomic gas to be constant, its specific heat at constant volume per mole (gram mole) is
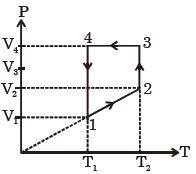
- 6A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 7The gas having average speed four times as that of $S{O_2}$ (molecular mass $64$) isView Solution
- 8View SolutionA piston is slowly pushed into a metal cylinder containing an ideal gas. Which of the following statements is/are incorrect?
- 9View SolutionThe ratio of mean kinetic energy of hydrogen and oxygen at a given temperature is
- 10A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
