Initial internal energy of the gas is ${U_1} = N\,\left( {\frac{5}{2}R} \right)\,T$
Since $n$ moles get dissociated into atoms, therefore, after heating, vessel contains $(N - n)$ moles of diatomic gas and $2n$ moles of a mono-atomic gas. Hence the internal energy for the gas, after heating, will be equal to
${U_2} = (N - n)\left( {\frac{5}{2}R} \right)\,T + 2n\,\left( {\frac{3}{2}R} \right)\,T$$ = \frac{5}{2}\,NRT + \,\frac{1}{2}nRT$
Hence, the heat supplied = increase in internal energy
$ = \,({U_2} - {U_1})\, = \,\frac{1}{2}\,nRT$
Download our appand get started for free
Similar Questions
- 1An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 2Heat is supplied to a diatomic gas at constant pressure. The ratio of $\Delta Q\,:\,\Delta U\,:\,\Delta W$ isView Solution
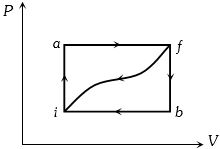
- 3When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
- 4Starting at temperature $300\; \mathrm{K},$ one mole of an ideal diatomic gas $(\gamma=1.4)$ is first compressed adiabatically from volume $\mathrm{V}_{1}$ to $\mathrm{V}_{2}=\frac{\mathrm{V}_{1}}{16} .$ It is then allowed to expand isobarically to volume $2 \mathrm{V}_{2} \cdot$ If all the processes are the quasi-static then the final temperature of the gas (in $\left. \mathrm{K}\right)$ is (to the nearest integer)View Solution
- 5$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
- 6A real gas within a closed chamber at $27^{\circ} \mathrm{C}$ undergoes the cyclic process as shown in figure. The gas obeys $P V^3=\mathrm{RT}$ equation for the path $A$ to $B$. The net work done in the complete cycle is (assuming $R=8 \mathrm{~J} / \mathrm{molK}$ ):View Solution
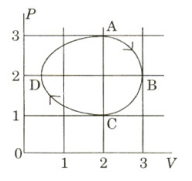
- 7The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA$. The part $ABC$ is a semi-circle and $CDA$ is half of an ellipse. Then,View Solution
$(A)$ the process during the path $\mathrm{A} \rightarrow \mathrm{B}$ is isothermal
$(B)$ heat flows out of the gas during the path $\mathrm{B} \rightarrow \mathrm{C} \rightarrow \mathrm{D}$
$(C)$ work done during the path $\mathrm{A} \rightarrow \mathrm{B} \rightarrow \mathrm{C}$ is zero
$(D)$ positive work is done by the gas in the cycle $ABCDA$
- 8View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
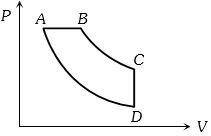
- 9View SolutionIn pressure-volume diagram given below, the isochoric, isothermal, and isobaric parts respectively, are
- 10A cylinder with a movable piston contains $3\,moles$ of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by having a pile of sand on it. By what factor does the pressure of the gas increases if the gas is compressed to half its original volume?View Solution
