A cylinder with a movable piston contains $3\,moles$ of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by having a pile of sand on it. By what factor does the pressure of the gas increases if the gas is compressed to half its original volume?
Easy
(a)
The process is adiabatic
$\therefore P _1 V _1^\gamma= P _2 V _2^\gamma$
Given, $\frac{ V _1}{ V _2}=\frac{2}{1}$ and $\gamma=\frac{7}{5}$ (for diatomic gas)
$\therefore \frac{ P _2}{ P _1}=(2)^{7 / 5} \quad \text { i.e., } P _2=(2)^{7 / 5} P _1$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
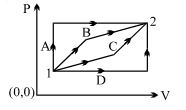
- 1View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
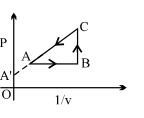
- 2An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 3$Assertion :$ In an isolated system the entropy increases.View Solution
$Reason :$ The processes in an isolated system are adiabatic. - 4View SolutionThe first law of thermodynamics is concerned with the conservation of
- 5During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?View Solution
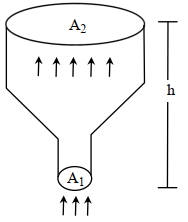
- 6An ideal gas of density $\rho=0.2 kg m ^{-3}$ enters a chimney of height $h$ at the rate of $\alpha=0.8 kg s ^{-1}$ from its lower end, and escapes through the upper end as shown in the figure. The cross-sectional area of the lower end is $A_1=0.1 m ^2$ and the upper end is $A_2=0.4 m ^2$. The pressure and the temperature of the gas at the lower end are $600 Pa$ and $300 K$, respectively, while its temperature at the upper end is $150 K$. The chimney is heat insulated so that the gas undergoes adiabatic expansion. Take $g=10 ms ^{-2}$ and the ratio of specific heats of the gas $\gamma=2$. Ignore atmospheric pressure.View Solution
Which of the following statement($s$) is(are) correct?
- 7An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
- 8A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
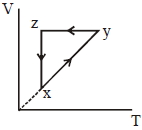
- 9A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
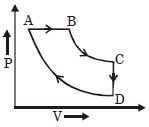
- 10Find $V_B = ?$View Solution