Starting at temperature $300\; \mathrm{K},$ one mole of an ideal diatomic gas $(\gamma=1.4)$ is first compressed adiabatically from volume $\mathrm{V}_{1}$ to $\mathrm{V}_{2}=\frac{\mathrm{V}_{1}}{16} .$ It is then allowed to expand isobarically to volume $2 \mathrm{V}_{2} \cdot$ If all the processes are the quasi-static then the final temperature of the gas (in $\left. \mathrm{K}\right)$ is (to the nearest integer)
JEE MAIN 2020, Diffcult
$\mathrm{PV} ^\gamma=$ constant
$\mathrm{TV} ^{\gamma-1}=\mathrm{C}$
$300 \times \mathrm{V}^{\frac{7}{5}-1}=\mathrm{T}_{2}\left(\frac{\mathrm{V}}{16}\right)^{\frac{7}{5}-1}$
$300 \times 2^{4 \times \frac{2}{5}}=\mathrm{T}_{2}$
Isobaric process
$\mathrm{V}=\frac{\mathrm{nRT}}{\mathrm{P}}$
$\mathrm{V}_{2}=\mathrm{kT}_{2}$
$2 \mathrm{V}_{2}=\mathrm{KT}_{\mathrm{f}}$
$\frac{1}{2}=\frac{\mathrm{T}_{2}}{\mathrm{T}_{\mathrm{f}}} \Rightarrow \mathrm{T}_{\mathrm{f}}=2 \mathrm{T}_{2}$
$\mathrm{T}_{\mathrm{f}}=2 \times 300 \times 2^{\frac{8}{5}}=1818.85$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
- 2Work done for the process shown in the figure is ............ $J$View Solution
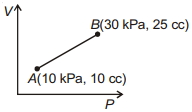
- 3The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.View Solution
- 4$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 5View SolutionIn a cyclic process, work done by the system is
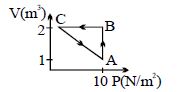
- 6An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
- 7A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 8View SolutionMonoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.
- 9View SolutionIn an isothermal change, an ideal gas obeys
- 10A gas is enclosed in a cylinder with a movable frictionless piston. Its initikl thermodynamic state at pressure $P_i=10^5 \mathrm{~Pa}$ and volume $V_i=10^{-3} \mathrm{~m}^3$ chanıes to i final state at $P_f=(1 / 32) \times 10^5 \mathrm{~Pa}$ and $V_f=8 \times 10^{-3} \mathrm{~m}^3$ in an adiabatic quasi-static process, such that $P^3 V^5=$ constant. Consider another thermodynamic process that brings the system from the same initial state to the same final state in two steps: an isobaric expansion at $P$, followed by an isochoric (isovolumetric) process at volume $V_f$. The amount of heat supplied to the system in the two-step process is approximatelyView Solution
