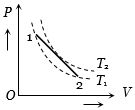
A volume $V$ and pressure $P$ diagram was obtained from state $1$ to state $2$ when a given mass of a gas is subjected to temperature changes. During this process the gas is

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The kinetic energy of translation of $20\, gm$ of oxygen at $47°C$ is (molecular wt. of oxygen is $32 \,gm/mol$ and $R = 8.3\, J/mol/K)$View Solution
- 2A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
- 3A gas at ${27}^\circ C$ temperature and $30$ atmospheric pressure is allowed to expand to the atmospheric pressure. If the volume becomes $10$ times its initial volume, then the final temperature becomes ...... $^oC$View Solution
- 4When unit mass of water boils to become steam at $100\,^0C$, it absorbs $Q$ amount of heat. The densities of water and steam at $100\,^0C$ are $\rho_1$ and $\rho_2$ respectively and the atmospheric pressure is $p_0$. The increase in internal energy of the water isView Solution
- 5At what temperature $r.m.s.$ speed of air molecules doubles of that at $N.T.P.$ is ...... $^oC$View Solution
- 6Consider a $1\, c.c.$ sample of air at absolute temperature ${T_0}$ at sea level and another $1 cc$ sample of air at a height where the pressure is one-third atmosphere. The absolute temperature $T$ of the sample at that height isView Solution
- 7The mean free path of gas molecules depends on $(d =$ molecular diameter$)$View Solution
- 8Consider a gas for which diameter of molecules is $\sigma $ , the gas is at a pressure $P$ and temperature $T$ and $N_a =$ avagodro’s number. The mean free path along $x$ axis isView Solution
- 9The temperature of a gas at pressure $P$ and volume $V$ is $27°C.$ Keeping its volume constant if its temperature is raised to $927°C,$ then its pressure will beView Solution
- 10The average distance between molecules of an ideal gas at $STP$ is approximately of the order ofView Solution