so, $\frac{{KA({\theta _1} - {\theta _2})t}}{l} = m \times L$
$ \Rightarrow m = \frac{{{{10}^{ - 3}} \times 92 \times (100 - 0) \times 60}}{{1 \times 8 \times {{10}^4}}} = 6.9 \times {10^{ - 3}}kg$
Download our appand get started for free
Similar Questions
- 1The rate of radiation of a black body at $0°C$ is $E\,J/sec$ . The rate of radiation of this black body at ${273^o}C$ will beView Solution
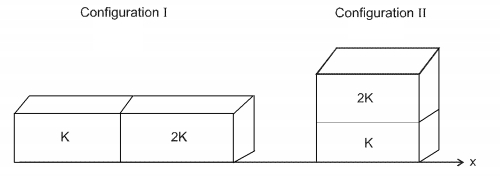
- 2Two rectangular blocks, having indentical dimensions, can be arranged either in configuration $I$ or in configuration $II$ as shown in the figure, On of the blocks has thermal conductivity $k$ and the other $2 \ k$. The temperature difference between the ends along the $x$-axis is the same in both the configurations. It takes $9\ s$ to transport a certain amount of heat from the hot end to the cold end in the configuration $I$. The time to transport the same amount of heat in the configuration $II$ is :View Solution
- 3View SolutionThe velocity of heat radiation in vacuum is
- 4Star $A$ has radius $ r$ surface temperature $T$ while star $B$ has radius $4r$ and surface temperature $T/2$ . The ratio of the power of two starts, $P_A : P_B$ isView Solution
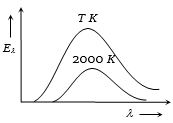
- 5The adjoining diagram shows the spectral energy density distribution ${E_\lambda }$of a black body at two different temperatures. If the areas under the curves are in the ratio $16 : 1$ , the value of temperature $T$ is ........ $K$View Solution
- 6$Assertion :$ It is hotter over the top of a fire than at the same distance of the sides.View Solution
$Reason :$ Air surrounding the fire conducts more heat upwards.
- 7View SolutionOn heating one end of a rod, the temperature of whole rod will be uniform when
- 8Read the following statements:View Solution
$A.$ When small temperature difference between a liquid and its surrounding is doubled the rate of loss of heat of the liquid becomes twice.
$B.$ Two bodies $P$ and $Q$ having equal surface areas are maintained at temperature $10^{\circ}\,C$ and $20^{\circ}\,C$. The thermal radiation emitted in a given time by $P$ and $Q$ are in the ratio $1: 1.15$
$C.$ A carnot Engine working between $100\,K$ and $400\,K$ has an efficiency of $75 \%$
$D.$ When small temperature difference between a liquid and its surrounding is quadrupled, the rate of loss of heat of the liquid becomes twice.
Choose the correct answer from the options given below :
- 9The intensity of radiation emitted by the sun has its maximum value at a wavelength of $510\;nm$ and that emitted by the north star has the maximum value at $350\;nm$. If these stars behave like black bodies, then the ratio of the surface temperature of the sun and north star isView Solution
- 10A black body radiates $ 20\,W$ at temperature ${227^o}C$. If temperature of the black body is changed to ${727^o}C$ then its radiating power will be ..... $W$View Solution
