Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
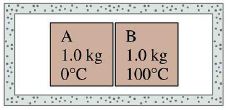
- 1Objects $A$ and $B$ that are initially separated from each other and well isolated from their surroundings are then brought into thermal contact. Initially $T_A= 0^oC$ and $T_B = 100^oC$. The specific heat of $A$ is less than the specific heat of $B$. After some time, the system comes to an equilibrium state. The final temperatures are :View Solution
- 2It takes $10$ minutes to cool a liquid from $61^oC$ to $59^oC$. If room temperature is $30^oC$ then time taken in cooling from $51^oC$ to $49^oC$ is ....... $\min$View Solution
- 3A solid sphere of radius $R$ and a hollow sphere of inner radius $r$ and outer radius $R$ made of copper are heated to the same temperature and are allowed to cool in the same environment. Then, choose the $CORRECT$ statementView Solution
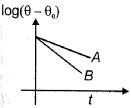
- 4Two bodies $A$ and $B$ of equal masses, area and emissivity cooling under Newton's law of cooling from same temperature are represented by the graph. If $\theta$ is the instantaneous temperature of the body and $\theta_0$ is the temperature of surroundings, then relationship between their specific heats is ..........View Solution
- 5Two identical beakers $A$ and $B$ contain equal volumes of two different liquids at $60\,^oC$ each and left to cool down. Liquid in $A$ has density of $8 \times10^2\, kg / m^3$ and specific heat of $2000\, Jkg^{-1}\,K^{-1}$ while liquid in $B$ has density of $10^3\,kgm^{-3}$ and specific heat of $4000\,JKg^{-1}\,K^{-1}$ . Which of the following best describes their temperature versus time graph schematically? (assume the emissivity of both the beakers to be the same)View Solution
- 6At $127^o C$ radiates energy is $2.7 \times 10^{-3} J/s$. At ....... $K$ temperature radiated energy is $4.32 \times 10^6 J/s$View Solution
- 7View SolutionThe dimensions of thermal resistance are
- 8View SolutionThe cause of Fraunhoffer lines is
- 9A beaker full of hot water is kept in a room. If it cools from $80^{\circ} C$ to $75^{\circ} C$ in $t_1$, minutes, from $75^{\circ} C$ to $70^{\circ} C$ in $t_2$ minutes and from $70^{\circ} C$ to $65^{\circ} C$ in $t_3$ minutes, thenView Solution
- 10Certain substance emits only the wavelengths ${\lambda _1},\;{\lambda _2},\;{\lambda _3}$ and ${\lambda _4}$ when it is at a high temperature. When this substance is at a colder temperature, it will absorb only the following wavelengthsView Solution
