$\mathrm{W}=\mathrm{n} \mathrm{RT} \log _{\mathrm{e}} \frac{\mathrm{V}_{2}}{\mathrm{V}_{1}}$
For, $n=1$
$\mathrm{W}=\mathrm{RT} \ln \frac{\mathrm{V}_{2}}{\mathrm{V}_{1}}$
For an isothermal process, $\Delta \mathrm{U}=0$ $\Delta \mathrm{Q}=\Delta \mathrm{U}+\mathrm{W}=0+\mathrm{RT} \ln \frac{\mathrm{V}_{2}}{\mathrm{V}_{1}}$
$\therefore$ Entropy $=\frac{\Delta Q}{T}=R \ln \frac{V_{2}}{V_{1}}$
Download our appand get started for free
Similar Questions
- 1View SolutionIn thermodynamics, heat and work are
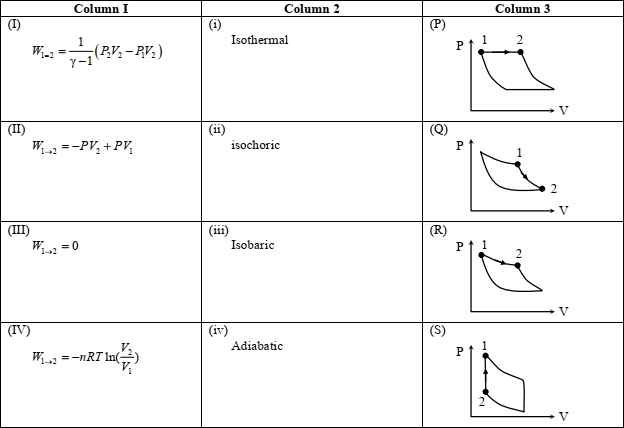
- 2An ideal gas is undergoing a cyclic thermodynamic process in different ways as shown in the corresponding $P$ $V$ diagrams in column $3$ of the table. Consider only the path from state $1$ to $2 . W$ denotes the corresponding work done on the system. The equations and plots in the table have standard notations as used in thermodynamic processes. Here $\gamma$ is the ratio of heat capacities at constant pressure and constant volume. The number of moles in the gas is $n$.View Solution
(image)
($1$) Which of the following options is the only correct representation of a process in which $\Delta U=\Delta Q-P \Delta V$ ?
$[A] (II) (iv) (R)$ $[B] (II) (iii) (P)$ $[C] (II) (iii) (S)$ $[D] (III) (iii) (P)$
($2$) Which one of the following options is the correct combination?
$[A] (III) (ii) (S)$ $[B] (II) (iv) (R)$ $[C] (II) (iv) (P)$ $[D] (IV) (ii) (S)$
($3$) Which one of the following options correctly represents a thermodynamic process that is used as a correction in the determination of the speed of sound in an ideal gas?
$[A] (III) (iv) (R)$ $[B] (I) (ii)$ $(\mathrm{Q})$ $[C] (IV) (ii) (R)$ $[D] (I) (iv) (Q)$
- 3In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
- 4Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
- 5View SolutionA gas is being compressed adiabatically. The specific heat of the gas during compression is
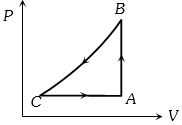
- 6A sample of an ideal gas is taken through a cycle a shown in figure. It absorbs $50J$ of energy during the process $AB$, no heat during $BC$, rejects $70J$ during $CA.$ $40J$ of work is done on the gas during $BC$. Internal energy of gas at $A$ is $1500J$, the internal energy at $C$ would be ........ $J$View Solution
- 7Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
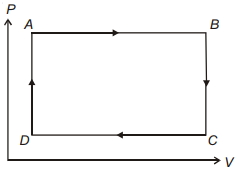
- 8The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
- 9Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 10A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
