In the process given, internal energy is
$U=a V^3$
$\Rightarrow \quad \frac{f n R T}{2} =a V^3$
where, $f=$ degree of freedom $=3$ (for ideal gas) and $n=$ number of moles $=1$.
$\Rightarrow \quad \frac{3}{2} R T=a V^3$
$\Rightarrow \quad \frac{3}{2} p V=a V^3 \quad[\because p V=R T]$
So, pressure in this process is given by
$p=\frac{2 a}{3} V^2$
Now, work done during the process is
$W=\int \limits_V^{2 V} p d V=\int \limits_V^{2 V} \frac{2 a}{3} V^2 d V$
$=\frac{2 a}{3} \times \frac{1}{3}\left((2 V)^3-V^3\right)$
$=\frac{2 a}{9}\left(7 V^3\right)$
$=\frac{2}{9} \times 7 \times \frac{3}{2} R T \quad\left[\because a V^3=\frac{3}{2} R^2 T\right]$
$=\frac{7}{3} R T$
Download our appand get started for free
Similar Questions
- 1A solid body of constant heat capacity $1\,J /{ }^{\circ} C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with 8 reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^{\circ}\,C$ to final temperature $200^{\circ}\,C$. Entropy change of the body in the two cases respectively is :
- 2Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
- 3The graph of pressure $(P)$ and volume $(V)$ according to $PV^n = C$, here $n = 1.4$View Solution
- 4A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
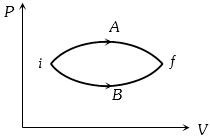
- 5In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
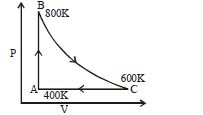
- 6One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 7A Carnot engine with sink's temperature at $17\,^oC$ has $50\%$ efficiency. By how much should its source temperature be changed to increases its efficiency to $60\%$ ?...... $K$View Solution
- 8Two moles of an ideal monoatomic gas at ${27^o}C$ occupies a volume of $V.$ If the gas is expanded adiabatically to the volume $2V,$ then the work done by the gas will be ....... $J$ $[\gamma = 5/3,\,R = 8.31J/mol\,K]$View Solution
- 9The pressure of an ideal gas varies with volume as $P = \alpha V,$ where $\alpha $ is a constant. One mole of the gas is allowed to undergo expansion such that its volume becomes $'m'$ times its initial volume. The work done by the gas in the process isView Solution
- 10A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
