A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$
JEE MAIN 2022, Medium
$\eta=1-\frac{ T _{ L }}{ T _{ H }}$
$\frac{1}{2}=1-\frac{ T _{ L }}{ T _{ H }}$
$\frac{1}{2}(1.3)=1-\left(\frac{ T _{ L }-40}{ T _{ H }}\right)$
$\frac{1}{2}(1.3)=\frac{1}{2}+\frac{40}{ T _{ H }} \quad T _{ H }=266.7\,K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
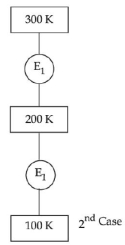
- 1In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 2An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 3A Carnot’s engine is made to work between $200°C$ and $0°C$ first and then between $0°C$ and $-200°C.$ The ratio of efficiencies of the engine in the two cases isView Solution
- 4Two kg of water is converted into steam by boiling at atmospheric pressure. The volume changes from $2 \times {10^{ - 3}}\,{m^3}$ to $3.34{m^3}.$ The work done by the system is about ....... $kJ$View Solution
- 5During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 6View SolutionThe process in which no heat enters or leaves the system is termed as
- 7$PV$ curve for the process whose $VT$ curve isView Solution
- 8One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 9View SolutionThe specific heat of a gas at constant pressure is more than that of the same gas at constant volume because
- 10A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution