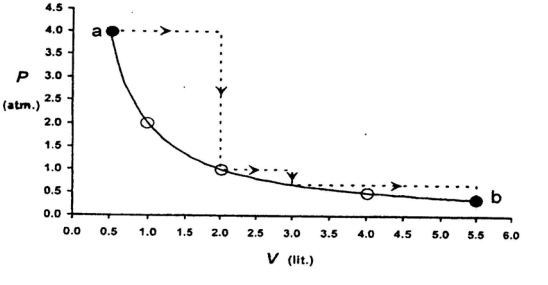
One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ is

IIT 2010, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$PV$ curve for the process whose $VT$ curve isView Solution
- 2In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correctView Solution
- 3View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 4During the adiabatic expansion of $2 \,moles$ of a gas, the internal energy was found to have decreased by $100 J$. The work done by the gas in this process is ..... $J$View Solution
- 5During an experiment, an ideal gas is found to obey a condition $VP^2 =$ constant. The gas is initially at a temperature $T$, pressure $P$ and volume $V.$ The gas expands to volume $4V$.View Solution
- 6A gas at initial temperature $T$ undergoes sudden expansion from volume $V$ to $2 \,V$. Then,View Solution
- 7A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
- 8A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
- 9View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
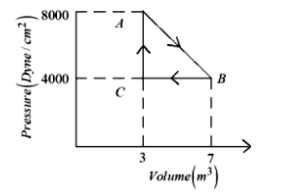
- 10A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution