One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will be
JEE MAIN 2018, Medium
$Work\,done\,on\,gas = nRT\,\ell n\left( {\frac{{{p_f}}}{{{p_l}}}} \right)$
$ = R\left( {300} \right)\ell n\left( 2 \right)\left( {\frac{{{p_f}}}{{{p_i}}} = 2\,given} \right)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
- 2A closed container contains a homogeneous mixture of two moles of an ideal monatomic gas $(\gamma=5 / 3)$ and one mole of an ideal diatomic gas $(\gamma=7 / 5)$. Here, $\gamma$ is the ratio of the specific heats at constant pressure and constant volume of an ideal gas. The gas mixture does a work of $66$ Joule when heated at constant pressure. The change in its internal energy is. . . . . . .Joule.View Solution
- 3View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
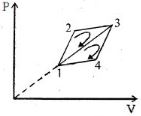
- 4The efficiency of a thermodynamic cycle $1-2-3- 1$ (see picture) is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta $ (in $\%$) of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution
- 5View SolutionFirst law of thermnodynamics is given by
- 6Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
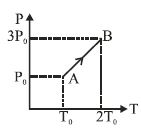
- 7Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
- 8One mole of an ideal gas at temperature $T_1$ expends according to the law $\frac{P}{{{V^2}}} =a$ (constant). The work done by the gas till temperature of gas becomes $T_2 $ isView Solution
- 9If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
- 10A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
