Download our appand get started for free
Similar Questions
- 1A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 2One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
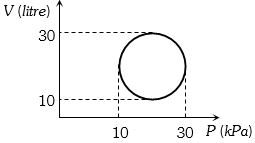
- 3View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
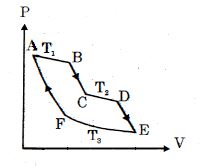
- 4One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas
- 5An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle isView Solution
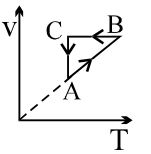
- 6A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 7If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 8One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
- 9One mole of an ideal gas at temperature $T_1$ expends according to the law $\frac{P}{{{V^2}}} =a$ (constant). The work done by the gas till temperature of gas becomes $T_2 $ isView Solution
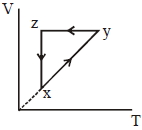
- 10View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?