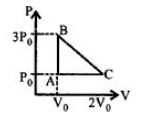
$p = {p_0} - \frac{{2{P_0}}}{{{V_0}}}\left( {V - 2{V_0}} \right)$
using=$PV=nRT$
Temperature,$T = \frac{{{P_0}V - \frac{{2{P_0}{V^2}}}{{{V_0}}} + 4{P_0}V}}{{1 \times R}}$
$\left( {n = 1\,mole\,given} \right)$
$T = \frac{{{P_0}}}{R}\left[ {5V - \frac{{2{V^2}}}{{{V_0}}}} \right]$
$\frac{{dT}}{{dV}} = 0 \Rightarrow 5 - \frac{{4V}}{{{V_0}}} = 0 \Rightarrow V = \frac{5}{4}{V_0}$
$T = \frac{{{P_0}}}{R}\left[ {5 \times \frac{{5{V_0}}}{4} - \frac{2}{{{V_0}}} \times \frac{{25}}{{16}}V_0^2} \right] = \frac{{25}}{8}\frac{{{P_0}{V_0}.}}{R}$
Download our appand get started for free
Similar Questions
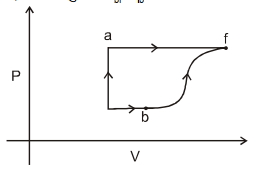
- 1A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
- 2View SolutionMonoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.
- 3A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 4Choose the incorrect statement from the followingView Solution
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
- 5The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
- 6$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
- 7Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 8For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
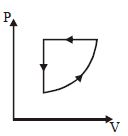
- 9View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
- 10An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. ThenView Solution
