Choose the incorrect statement from the following
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
Medium
Efficiency of heat engine, $\eta=1-\frac{\mathrm{T}_{2}}{\mathrm{T}_{1}}$
For $\eta=1 \text { (i.e. } 100 \%)$ either, $\mathrm{T}_{1}=\infty$ or $\mathrm{T}_{2}=0 \mathrm{K}$
As source at infinite temperature or sink at $0\, \mathrm{K}$ are not attainable.
Therefore heat engine cannot have efficiency $1$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
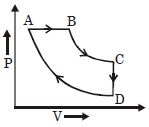
- 2A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
- 3$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
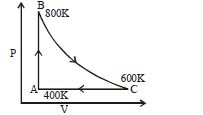
$Reason :$ Entropy increases in all natural processes. - 4One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
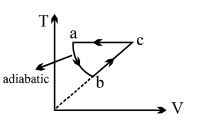
- 5$PV$ curve for the process whose $VT$ curve isView Solution
- 6Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
- 7If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
- 8Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
- 9A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 10View SolutionDuring the thermodynamic process shown in figure for an ideal gas