Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$
JEE MAIN 2018, Diffcult
Efficiency of engine $A,{n_A} = \frac{{{T_1} - {T_2}}}{{{T_1}}}$
and ${n_B} = \frac{{{T_2} - {T_3}}}{{{T_2}}};{T_2} = \frac{{{T_1} + {T_3}}}{2} = 350\,K$
$or\frac{{{n_A}}}{{{n_B}}} = \frac{{\frac{{600 - 350}}{{600}}}}{{\frac{{350 - 100}}{{350}}}} = \frac{7}{{12}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermo-dynamical system is changed from state $({P_1},\,{V_1})$ to $({P_2},\,{V_2})$ by two different process. The quantity which will remain same will beView Solution
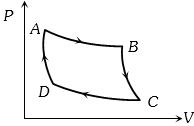
- 2Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
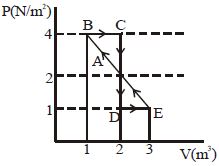
- 3One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
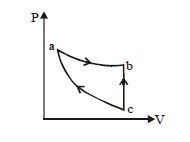
- 4View SolutionIrreversible process is
- 5A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
- 6An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 7Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$View Solution
- 8An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
- 9When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 10An ideal Carnot heat engine with an efficiency of $30\%$.It absorbs heat from a hot reservoir at $727^o C$. The temperature of the cold reservoir is .... $^oC$View Solution
