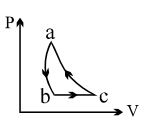
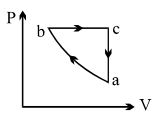
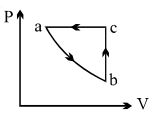
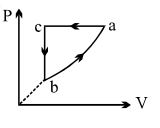
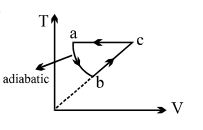
$PV$ curve for the process whose $VT$ curve is

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
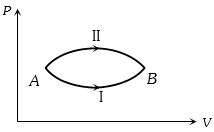
- 1A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
- 2If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 3Initial pressure and volume of a gas are $ P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $ V$. The final pressure of gas will beView Solution
- 4Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
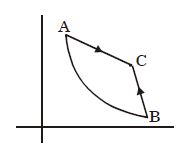
- 5Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
- 6A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
- 7The pressure in the tyre of a car is four times the atmospheric pressure at $300 K$. If this tyre suddenly bursts, its new temperature will be $(\gamma = 1.4)$View Solution
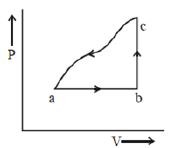
- 8A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 9The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution
- 10A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution