==>$\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{P_1}}}{{{P_2}}}} \right)^{\frac{{1 - \gamma }}{\gamma }}}$
==>$\frac{{{T_2}}}{{300}} = {\left( {\frac{4}{1}} \right)^{\frac{{(1 - 1.4)}}{{1.4}}}}$
==>${T_2} = 300{(4)^{ - \frac{{0.4}}{{1.4}}}}$
Download our appand get started for free
Similar Questions
- 1The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution
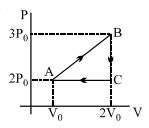
- 2A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
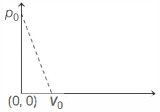
- 3One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
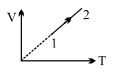
- 4An ideal gas undergoes the process $1 \rightarrow 2$ as shown in the figure, the heat supplied and work done in the process is $\Delta \,\,Q$ and $\Delta \,\,W$ respectively. The ratio $\Delta \,\,Q :$ $\Delta \,\,W$ isView Solution
- 5A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
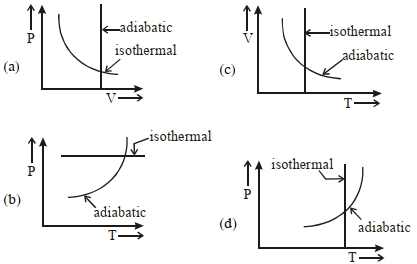
- 6View SolutionWhich one is the correct option for the two different thermodynamic processes ?
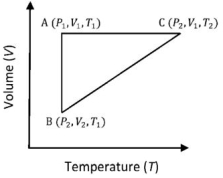
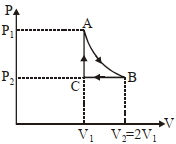
- 7$n$ mole a perfect gas undergoes a cyclic process $ABCA$ (see figure) consisting of the following processes.View Solution
$A \rightarrow B :$ Isothermal expansion at temperature $T$ so that the volume is doubled from $V _{1}$ to $V _{2}=2 V _{1}$ and pressure changes from $P _{1}$ to $P _{2}$
$B \rightarrow C :$ Isobaric compression at pressure $P _{2}$ to initial volume $V _{1}$
$C \rightarrow A$ : Isochoric change leading to change of pressure from $P _{2}$ to $P _{1}$
Total workdone in the complete cycle $ABCA$ is
- 8View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
- 9View SolutionWhich is the correct statement
- 10View SolutionIn the above thermodynamic process, the correct statement is