Initial pressure and volume of a gas are $ P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $ V$. The final pressure of gas will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIf a cylinder containing a gas at high pressure explodes, the gas undergoes
- 2The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 3View SolutionIn Carnot engine the work done by working substance is equivalent to
- 4Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
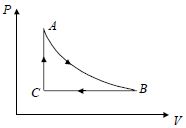
- 5A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
- 6View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
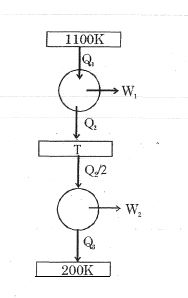
- 7Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 8A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
- 9A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 10An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
