$P _{ atm }= P cm$ of water
What we can conclude from this process is that the volume is changing in the air bubble but the temperature remains unchanged.
For isothermal process,
$P _{1} V _{1}= P _{2} V _{2}$
Let the height of water surface be $x$.
$(P d g+x d g)\left(\frac{4}{3} \pi r^{3}\right)=P d g\left[\frac{4}{3} \pi(2 r)^{3}\right]$
$(P+x) r^{3}=P\left(8 r^{3}\right)$
$x=8 P-P$
$\Rightarrow x=7 P$
Download our appand get started for free
Similar Questions
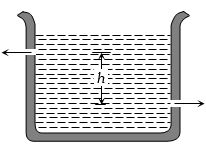
- 1There are two identical small holes of area of cross-section a on the opposite sides of a tank containing a liquid of density $\rho $. The difference in height between the holes is $h$. Tank is resting on a smooth horizontal surface. Horizontal force which will has to be applied on the tank to keep it in equilibrium isView Solution
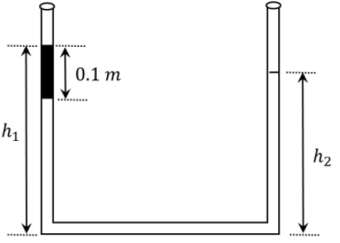
- 2An open-ended U-tube of uniform cross-sectional area contains water (density $10^3 kg m ^{-3}$ ). Initially the water level stands at $0.29 m$ from the bottom in each arm. Kerosene oil (a water-immiscible liquid) of density $800 kg m ^{-3}$ is added to the left arm until its length is $0.1 m$, as shown in the schematic figure below. The ratio $\left(\frac{h_1}{h_2}\right)$ of the heights of the liquid in the two arms is-View Solution
- 3The water is filled upto height of $12\, {m}$ in a tank having vertical sidewalls. A hole is made in one of the walls at a depth $'{h}'$ below the water level. The value of $'h'$ for which the emerging stream of water strikes the ground at the maximum range is $.....\,{m}$.View Solution
- 4If $\rho$ is the density and $\eta$ is coefficient of viscosity of fluid which flows with a speed $v$ in the pipe of diameter $d$, the correct formula for Reynolds number $R _{ e }$ is ..............View Solution
- 5View SolutionIn Poiseuilli's method of determination of coefficient of viscosity, the physical quantity that requires greater accuracy in measurement is
- 6Two identical cylindrical vessels are kept on the ground and each contain the same liquid of density $d.$ The area of the base of both vessels is $S$ but the height of liquid in one vessel is $x_{1}$ and in the other, $x_{2}$. When both cylinders are connected through a pipe of negligible volume very close to the bottom, the liquid flows from one vessel to the other until it comes to equilibrium at a new height. The change in energy of the system in the process isView Solution
- 7A capillary tube is attached horizontally to a constant head arrangement. If the radius of the capillary tube is increased by $10\%$ then the rate of flow of liquid will change nearly by ......... $\%$View Solution
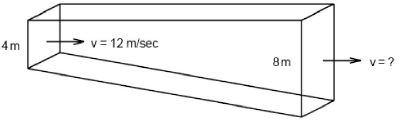
- 8A river gradually deepens, from a depth of $4 \ m$ to a depth of $8\ m$ as shown. The width, $W$, of the river does not change. At the depth of $4 \ m$, the river's speed is $12\ m/sec.$ Its elocity at the $8\ m$ depth is ......... $m/sec$View Solution
- 9$A U-$ tube having horizontal arm of length $20$ $cm$, has uniform cross-sectional area $=1\ cm^2$. It is filled with water of volume $60$ $cc$. What volume of a liquid of density $4$ $g/cc$ should be poured from one side into the $U -$ tube so that no water is left in the horizontal arm of the tube ........ $cc$ ?View Solution
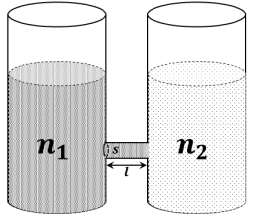
- 10As shown schematically in the figure, two vessels contain water solutions (at temperature $T$ ) of potassium permanganate $\left( KMnO _4\right)$ of different concentrations $n_1$ and $n_2\left(n_1>n_2\right)$ molecules per unit volume with $\Delta n=\left(n_1-n_2\right) \ll n_1$. When they are connected by a tube of small length $\ell$ and cross-sectional area $S , KMnO _4$ starts to diffuse from the left to the right vessel through the tube. Consider the collection of molecules to behave as dilute ideal gases and the difference in their partial pressure in the two vessels causing the diffusion. The speed $v$ of the molecules is limited by the viscous force $-\beta v$ on each molecule, where $\beta$ is a constant. Neglecting all terms of the order $(\Delta n)^2$, which of the following is/are correct? ( $k_B$ is the Boltzmann constant)-View Solution
$(A)$ the force causing the molecules to move across the tube is $\Delta n k_B T S$
$(B)$ force balance implies $n_1 \beta v \ell=\Delta n k_B T$
$(C)$ total number of molecules going across the tube per sec is $\left(\frac{\Delta n}{\ell}\right)\left(\frac{k_B T}{\beta}\right) S$
$(D)$ rate of molecules getting transferred through the tube does not change with time