For free expansion, \(W=0\) and for adiabatic process, \(q=0\).
\(\because \Delta U=q+W=0\) this means that internal energy remains constant. Therefore, \(\Delta T=0\) in ideal gas as there is no intermolecular attraction. Hence, when such a gas expands under adiabatic conditions into a vacuum, no heat is absorbed or evolved. Since, no external work is done to separate the molecules.
Download our appand get started for free
Similar Questions
- 1$25^{\circ} C$ અને $1$ વાતા દબાણે દહન એન્થાલ્પીનો નીયે આપેલ છે.View Solution
પદાર્થ $H _{2}$ $C$(ગ્રેફાઈટ) $C _{2} H _{6}( g )$ $\frac{\Delta_{ C } H ^{\Theta}}{ kJmol ^{-1}}$ $-286.0$ $-394.0$ $-1560.0$ તો,ઈથેનની સર્જન એન્થાલ્પી ........
- 2નીચેની માહિતી પરથી નાઇટ્રિક ઓક્સાઇડની સર્જન એન્થાલ્પી કેટલા .......$kJ\,mo{l^{ - 1}}$ થશે ?$N{O_{\left( g \right)}} + C{O_{\left( g \right)}} \to 1/2{N_{2\left( g \right)}} + C{O_{2\left( g \right)}};\Delta {H^o} = - 372.2\,kJ/mol$View Solution
$\Delta H_f^o\left( {CO} \right) = - 110.5\,kJ\,mo{l^{ - 1}};$
$\Delta H_f^o\left( {C{O_2}} \right) = - 393.5\,kJ\,mo{l^{ - 1}}$
- 3$H_2 + 1/2 O_2$ $\rightarrow$ $H_2O$ : $\Delta H = -68.4$ કિલોજૂલ, $C + O_2$ $\rightarrow$ $CO_2$ : $\Delta H = -94.0$ કિલોકેલરી અને $C_2H_5OH + 3O_2 $$\rightarrow$ $2CO_2 + 3H_2O$ : $\Delta H = -327.0$ કિલોકેલરી, તો $C_2H_5OH$ ની સર્જન ઉષ્મા $= ......$ કિલોકેલરીView Solution
- 4$298$ કેલ્વિને $2$ મોલ આદર્શ વાયુનું $10$ લિટરથી $20$ લિટરમાં પ્રતિવર્ષી સમઉષ્મીય વિસ્તરણ દરમિયાન થતાં કાર્યની ગણતરી .....$J$ થશે.View Solution
- 5અચળ તાપમાને અને દબાણે થતી નીચેની પ્રક્રિયા ધ્યાનમાં લો.View Solution
${N_2} + 3{H_2} \to 2N{H_3}$
જો $\Delta H$ અને $\Delta U$ અનુક્રમે પ્રક્રિયા માટેના એન્થાલ્પી ફેરફાર અને આંતરિક ઊર્જા ફેરફાર હોય, તો નીચેનામાંથી કઇ રજૂઆત સાચી છે ?
- 6$100^{\circ} {C}$ અને $1\, bar$એ પાણી માટેView Solution
$\Delta_{\text {vap }} {H}-\Delta_{\text {vap }} {U}=...... \times 10^{2} \,{~J}\, {~mol}^{-1}$.
$\left[\right.$ ઉપયોગ કરો : $\left.R=8.31\, {~J}\, {~mol}^{-1}\, {~K}^{-1}\right]$
[${H}_{2} {O}({l})$નું કદ ${H}_{2} {O}({g})$ના કદ કરતાં ઘણું નાનું ધારો. ધારો કે ${H}_{2} {O}({g})$ને આદર્શ વાયુ તરીકે ગણવામાં આવે છે]
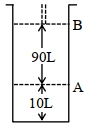
- 7આપેલ આક્રૂતિને ધ્યાનમાં લો.View Solution
$18^{\circ} \mathrm{C}$ પર, સ્થાન $A$ પર, પિસ્ટન સાથે જોડેલા (fitted) સિલિન્ડર માં આદર્શ વાયુનો $1$ $\mathrm{mol}$ રાખેલ છે. જો તાપમાન માં કોઈપણ જાતનો ફેરફાર ન કરીએ તો પિસ્ટન એ સ્થાન $B$ તરફ ખસે છે ત્યારે આ પ્રતિવર્તી પ્રક્રમ માં થયેલ કાર્ય $'x' L atm$ છે. $x=-$ ........... $L.atm$ (નજીક નો પૂર્ણાક)
[આપેલ : નિરપેક્ષ તાપમાન $={ }^{\circ} \mathrm{C}+273.15, \mathrm{R}=0.08206 \mathrm{~L} \mathrm{~atm} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ ]
- 8અચળ તાપમાને અને દબાણે પ્રણાલી માટે ગીબ્સ ઉર્જાનો ફેરફાર ($\Delta G$ પ્રણાલી ) માટે સાચું વિધાન ઓળખો.View Solution
- 9મિથેનના દહનનું $\Delta U^{\theta}$ મૂલ્ય $-X \,kJ \,mol ^{-1}$ છે. $\Delta H^{\theta}$ નું મૂલ્ય શું હશે ?View Solution
- 10View Solutionએન્ટ્રોપી એટલે .......
