$V_1 = 2\, litre$, $V_2 = 2.5$ લીટર , $ q = + 300, Pext = 1$ વાતાવરણ
તેથી, $\Delta U = q + W$
$W = -P_{ext} \Delta V = -P_{ext} (V_2 - V_1) = -P_{ext}(2.5 - 2) = -1 (0.5) = -0.5 \,lit. atm$
નોંધ: $1\, lit - atm = 101.3\, Joule, 1\, Calorie = 4.2\, Joule$
તેથી,$W = -0.5 × 101.3 = -50.65$ જુલ
$\Delta U = q + W = 300 + (-50.65) = 249.35 \,J$
Download our appand get started for free
Similar Questions
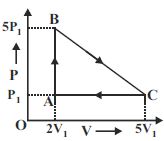
- 1View Solutionએક આદર્શ વાયુ નીચે મુજબ પ્રતિવર્તી ચકીય પ્રકમ અનુભવે તો આ પ્રકમમાં થતુ કાર્ય ........
- 2View Solutionસ્વયંભૂ પ્રક્રિયા થવા માટે.......
- 3$X_2$, $Y_2$ અને $XY_3$ ના પ્રમાણિત એન્ટ્રોપી અનુક્રમે $60, 40$ અને $50 $ $J\,K^{-1}$ $mol^{-1}$ છે. તો, $\frac{1}{2}{X_2}\, + \,\,\frac{3}{2}{Y_2}\, \to \,X{Y_3},\,\Delta H\,\, = \,\, - 30\,\,kJ,$ પ્રક્રિયાના સંતુલન વખતે કેટલા ......$K$ તાપમાન હશે ?View Solution
- 4નીચે આપેલ પરિબળો દ્વારા જલીય દ્રાવણમાં ક્લોરીનની ઓક્સિડાઈઝ ક્ષમતા માપી શકાય છે. તો $1/2 Cl_2$$_{(g)}$ થી $Cl^{-}$ $_{(aq)}$માં રૂપાંતરણ થવા કેટલા .....$kJ\, mol^{-1}$ ઉર્જા ભાગ લેશે ?View Solution
$\frac{1}{2}C{l_2}_{(g)}\,\xrightarrow{{\frac{1}{2}{\Delta _{diss}}{H^\Theta }}}\,Cl_{(g)}\,\,\xrightarrow{{{\Delta _{eg}}{H^\Theta }}}\,\,C{l^ - }_{(g)}\,\xrightarrow{{{\Delta _{hyd}}{H^\Theta }}}\,C{l^ - }_{(aq)}$
$({\mkern 1mu} {\Delta _{diss}}{\mkern 1mu} H_{C{l_2}}^\Theta {\mkern 1mu} = {\mkern 1mu} {\mkern 1mu} 240{\mkern 1mu} {\mkern 1mu} kJ{\mkern 1mu} {\mkern 1mu} mo{l^{ - 1}},{\mkern 1mu} {\mkern 1mu} {\Delta _{eg}}{\mkern 1mu} H_{Cl}^\Theta {\mkern 1mu} = {\mkern 1mu} {\mkern 1mu} - 349{\mkern 1mu} {\mkern 1mu} kJ{\mkern 1mu} {\mkern 1mu} mo{l^{ - 1}},{\mkern 1mu} {\mkern 1mu} $
${\Delta _{hyd}}H_{C{l^ - }}^\Theta {\mkern 1mu} = {\mkern 1mu} {\mkern 1mu} - {\mkern 1mu} 381{\mkern 1mu} kJ{\mkern 1mu} {\mkern 1mu} mo{l^{ - 1}})$
- 5$T$ તાપમાને થતી એક પ્રતિવર્તી પ્રક્રિયા માટે $\Delta H$ અને $\Delta S$ બંને $+ve$ છે. જો સંતુલન સમયનું તાપમાન $T_e$ હોય તો આ પ્રક્રિયા સ્વયંભૂ ત્યારે બનશે જ્યારે ....View Solution
- 6View Solutionબોમ્બ કેલરીમીટરમાં પ્રક્રિયા માટે માપવામાં આવતી ઉષ્માનું પ્રમાણ છે?
- 7$2Zn + O_2$ $\rightarrow$ $2ZnO, \Delta G^o = -616 \,J ; 2Zn + S_2$ $\rightarrow$ $2ZnS$, $\Delta G^o = -293\, J; S_2 + 2O_2$ $\rightarrow$ $2SO_2$, $\Delta G^o = -408 \,J$View Solution
તો $2ZnS + 3O_2$ $\rightarrow$ $2ZnO + 2SO_3$ પ્રક્રિયા માટે $\Delta\, G^o$ નું મૂલ્ય .......$J$
- 8પ્રકિયા $3F{e_{\left( s \right)}} + 4{H_2}{O_{\left( g \right)}} \rightleftharpoons F{e_3}{O_{4\left( s \right)}} + 4{H_{2\left( g \right)}}$ એ પ્રતિવર્તી ત્યારે થશે જ્યારે તે ................ કરવામાં આવે.View Solution
- 9સ્પ્રિંગના સંકોચન દરમિયાન $10\,kJ$ કાર્ય થાય છે અને $2\,kJ$ ઉષ્મા તરીકે પર્યાવરણમાં મુક્ત થાય છે. તો આંતરિક ઊર્જામાં થતો ફેરફાર $\Delta U$ ($kJ$ માં) જણાવો.View Solution
- 10આપેલ બંધઊર્જાના મૂલ્યો પરથી નીચેની પ્રક્રિયાની એન્થાલ્પી કેટલા ................ $\mathrm{kJ \,mol}^{-1}$ થશે ?View Solution
$H - H$ બંધઊર્જા $:\, 431.37 \,kJ\, mol^{-1}$ $C= C$ બંધઊર્જા $:\, 606.10\, kJ \,mol^{-1}$ $C - C$ બંધઊર્જા $:\, 336.49\, kJ\, mol^{-1}$ $C - H$ બંધઊર્જા $:\, 410.50\, kJ\, mol^{-1}$ પ્રક્રિયા : $\begin{array}{*{20}{c}}
{H\,\,\,\,H} \\
{|\,\,\,\,\,\,\,\,|} \\
{C = C} \\
{|\,\,\,\,\,\,\,\,\,|} \\
{H\,\,\,\,H}
\end{array}\, + \,H - H\, \to \,\begin{array}{*{20}{c}}
{H\,\,\,\,H} \\
{|\,\,\,\,\,\,\,\,|} \\
{H - C - C - H} \\
{|\,\,\,\,\,\,\,\,\,|} \\
{H\,\,\,\,H}
\end{array}\,$
