The average kinetic energy of a gas at $-23°C$ and $75\, cm$ pressure is $5 \times {10^{ - 14}}\,erg$ for ${H_2}$. The mean kinetic energy of the ${O_2}$ at $227°C$ and $150\, cm$ pressure will be
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of a gas having $2.0 \times 10^{25}$ molecules per cubic meter at $1.38 \mathrm{~atm}$ (Given, $\mathrm{k}=$ $\left.1.38 \times 10^{-23} \mathrm{JK}^{-1}\right)$ is :View Solution
- 2View SolutionOn colliding in a closed container the gas molecules
- 3The total momentum of the molecules of $1 \,gm$ $mol$ of a gas in a container at rest of $300 \,K$ isView Solution
- 4View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
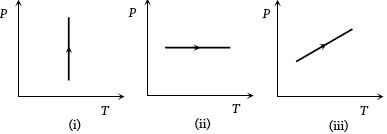
- 5An ideal gas is enclosed in a cylinder at pressure of $2\,atm$ and temperature, $300\,K.$ The mean time between two successive collisions is $6 \times 10^{-8}\, s.$ If the pressure is doubled and temperature is increased to $500\,K,$ the mean time between two successive collisions will be close toView Solution
- 6Consider a $1\, c.c.$ sample of air at absolute temperature ${T_0}$ at sea level and another $1 cc$ sample of air at a height where the pressure is one-third atmosphere. The absolute temperature $T$ of the sample at that height isView Solution
- 7$125\, ml$ of gas $A$ at $0.60$ atmosphere and $150\, ml$ of gas $B$ at $0.80$ atmosphere pressure at same temperature is filled in a vessel of $1$ litre volume. What will be the total pressure of mixture at the same temperature ............... $\mathrm{atmosphere}$View Solution
- 8One kg of a diatomic gas is at a pressure of $8 \times 10^4$ $N/m^2$ The density of the gas is $4$ $kg/m^3$ What is the energy (in $\times 10^4\; J$) of the gas due to its thermal motion?View Solution
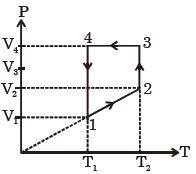
- 9A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 10If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
