$125\, ml$ of gas $A$ at $0.60$ atmosphere and $150\, ml$ of gas $B$ at $0.80$ atmosphere pressure at same temperature is filled in a vessel of $1$ litre volume. What will be the total pressure of mixture at the same temperature ............... $\mathrm{atmosphere}$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe parameter that remains the same for molecules of all gases at a given temperature is :
- 2The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?View Solution
- 3A closed cylindrical vessel contains $N$ moles of an ideal diatomic gas at a temperature $T$. On supplying heat, temperature remains same, but $n$ moles get dissociated into atoms. The heat supplied is .........View Solution
- 4The temperature $(T)$ of one mole of an ideal gas varies with its volume $(V)$ as $T=-\alpha V^3+\beta V^2$, where $\alpha$ and $\beta$ are positive constants. The maximum pressure of gas during this process is ............View Solution
- 5When the temperature of a gas is raised from $27^o C$ to $90^o C$, the percentage increase in the $r.m.s.$ velocity of the molecules will be ..... $\%$View Solution
- 6A gas mixture consists of molecules of type $1, 2$ and $3$, with molar masses ${m_1} > {m_2} > {m_3}.$ ${V_{rms}}$ and $\overline K $ are the $r.m.s.$ speed and average kinetic energy of the gases. Which of the following is trueView Solution
- 7Two vessels of the same volume contain the same gas at same temperature. If the pressure in the vessels be in the ratio of $1 : 2$, thenView Solution
- 8An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $P$ and volume $V$ is given by $PV^n =$ constant, then n is given by (Here $C_p$ and $C_v$ are molar specific heat at constant pressure and constant volume, respectively) :View Solution
- 9A flask contains Hydrogen and Argon in the ratio $2: 1$ by mass. The temperature of the mixture is $30^{\circ} C$. The ratio of average kinetic energy per molecule of the two gases ( $K$ argon/ $K$ hydrogen) is: (Given: Atomic Weight of $Ar = 39.9$)View Solution
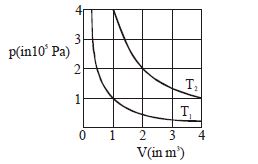
- 10The given diagram shows isotherms for a fixed mass of an ideal gas at temperature $T_1$ and $T_2$ . What is the value of the ratio $\frac{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_{2{\text{ }}}}}}{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_1}}}$ ?View Solution