The average kinetic energy of a molecule of the gas is
JEE MAIN 2023, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$5$ moles of oxygen is heated at constant volume from $10°C$ to $20°C.$ The change in the internal energy of the gas is (the gram molecular specific heat of oxygen at constant pressure...... $cal$ ${C_p} = 8$ $cal/mole \,°C$ and $R = 8.3 cal/mole\, °C)$View Solution
- 2A gas at absolute temperature $300\,K$ has pressure $= 4 \times 10^{-10}\,N /m^2$ . Boltzmann constant, $k = 1.38 \times 10^{-23}\,J / K$ . The number of molecules per $cm^3$ is of the order ofView Solution
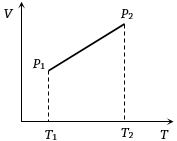
- 3From the following $V-T$ diagram we can concludeView Solution
- 4The energy (in $eV$) possessed by a neon atom at $27^{\circ} C$ is ..............View Solution
- 5View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 6Two ideal gases at absolute temperature $T_1$ and $T_2$ are mixed. There is no loss of energy. The masses of the molecules are $m_1$ and $m_2$ and the number of molecules in the gases are $n_1$ and $n_2$ respectively. The temperature of mixture will beView Solution
- 7When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 8The $r.m.s.$ velocity will be greater forView Solution
- 9One kg of a diatomic gas is at a pressure of $8 \times 10^4$ $N/m^2$ The density of the gas is $4$ $kg/m^3$ What is the energy (in $\times 10^4\; J$) of the gas due to its thermal motion?View Solution
- 10Consider two ideal diatomic gases $\mathrm{A}$ and $\mathrm{B}$ at some temperature $T$. Molecules of the gas $A$ are rigid, and have a mass $m$. Molecules of the gas $\mathrm{B}$ have an additional vibrational mode, and have a mass $\frac{\mathrm{m}}{4} .$ The ratio of the specific heats $(\mathrm{C}_{\mathrm{v}}^{\mathrm{A}}$ and $\mathrm{C}_{\mathrm{v}}^{\mathrm{B}})$ of gas $\mathrm{A}$ and $\mathrm{B}$, respectively isView Solution
