From the following $V-T$ diagram we can conclude
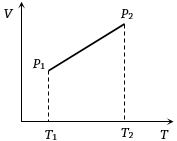

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
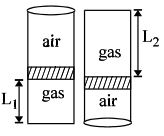
- 1An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$View Solution
- 2View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
- 3Let $\bar v,\;{v_{rms}}$ and ${v_p}$ respectively denote the mean speed, root mean square speed and most probable speed of the molecules in an ideal monoatomic gas at absolute temperature $T.$ The mass of a molecule is $m.$ ThenView Solution
- 4An ideal gas has an initial pressure of $3$ pressure units and an initial volume of $4$ volume units. The table gives the final pressure and volume of the gas (in those same units) in four, processes. Which processes start and end on the same isothermView Solution
$\begin{array}{|c|c|c|c|c|} \hline & A & B & C & D \\ \hline P & 5 & 4 & 12 & 6 \\ \hline V & 7 & 6 & 1 & 3 \\ \hline \end{array}$
- 5The curve between absolute temperature and ${v^2}_{rms}$ isView Solution
- 6When temperature of an ideal gas is increased from $27^\circ C$ to $227^\circ C$, its $r.m.s.$ speed changed from $400\, metre/sec$ to ${V_s}.$ The ${V_s}$ is ........ $metre/sec$View Solution
- 7View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
- 8An ideal gas initially at pressure $1$ bar is being compressed from $30 \,m ^{3}$ to $10\, m ^{3}$ volume and its temperature decreases from $320\, K$ to $280\, K$. then find final pressure of gas (in bar)View Solution
- 9$P-V$ diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will beView Solution
- 10View SolutionWhich of the following statements about kinetic theory of gases is wrong
