A gas at absolute temperature $300\,K$ has pressure $= 4 \times 10^{-10}\,N /m^2$ . Boltzmann constant, $k = 1.38 \times 10^{-23}\,J / K$ . The number of molecules per $cm^3$ is of the order of
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
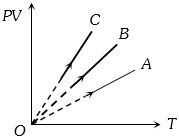
- 1$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
- 2A closed cylindrical vessel contains $N$ moles of an ideal diatomic gas at a temperature $T$. On supplying heat, temperature remains same, but $n$ moles get dissociated into atoms. The heat supplied is .........View Solution
- 3View SolutionWhich one the following graphs represents the behaviour of an ideal gas
- 4The molecules of a given mass of a gas have $r.m.s.$ velocity of $200 \,m s^{-1}$ at $27^o C$ and $1.0 \times 10^5 \,Nm^{-2}$ pressure. When the temperature and pressure of the gas are respectively, $127^o C$ and $0.05 \times 10^5 \,Nm^{-2},$ the $r.m.s.$ velocity of its molecules in $m s^{-1}$ isView Solution
- 5A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 6In an ideal gas at temperature $T$, the average force that a molecule applies on the walls of a closed container depends on $T$ as ${T^q }$ . A good estimate for $q$ isView Solution
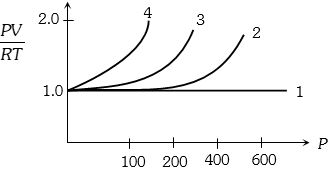
- 7A fix amount of nitrogen gas ($1$ mole) is taken and is subjected to pressure and temperature variation. The experiment is performed at high pressure as well as high temperatures. The results obtained are shown in the figures. The correct variation of $PV/RT$ with $P$ will be exhibited byView Solution
- 8Under which of the following conditions is the law $PV = RT$ obeyed most closely by a real gasView Solution
- 9View SolutionFor an ideal gas
- 10If the intermolecular forces vanish away, the volume occupied by the molecules contained in $4.5 \,kg$ water at standard temperature and pressure will beView Solution
