The average kinetic energy of a monatomic molecule is $0.414 \mathrm{eV}$ at temperature :
(Use $\mathrm{K}_{\mathrm{B}}=1.38 \times 10^{-23} \mathrm{~J} / \mathrm{mol}-\mathrm{K}$ )
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
- 2Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$) - 3View SolutionSelect the incorrect statement about ideal gas.
- 4$7$ mole of certain monoatomic ideal gas undergoes a temperature increase of $40 K$ at constant pressure. The increase in the internal energy of the gas in this process is$....J$ (Given $R =8.3 JK ^{-1} mol ^{-1}$ )View Solution
- 5At a given temperature, the pressure of an ideal gas of density $\rho $ is proportional toView Solution
- 6Two thermodynamical process are shown in the figure. The molar heat capacity for process $A$ and $B$ are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.View Solution
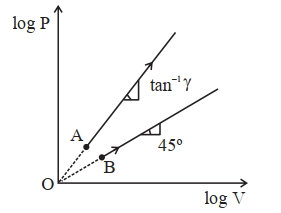
- 7Two ideal gases at absolute temperature $T_1$ and $T_2$ are mixed. There is no loss of energy. The masses of the molecules are $m_1$ and $m_2$ and the number of molecules in the gases are $n_1$ and $n_2$ respectively. The temperature of mixture will beView Solution
- 8The temperature of an ideal gas is increased from $120\, K$ to $480\, K.$ If at $120\, K,$ the root mean square velocity of the gas molecules is $v,$ at $480\, K$ it becomesView Solution
- 9The average speed $v$ and $r.m.s.$ speed $\bar v$ of the molecules are related asView Solution
- 10View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
