Select the incorrect statement about ideal gas.
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
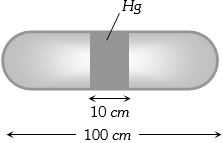
- 1A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 2When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 3For a gas $\frac{R}{{{C_V}}} = 0.67$. This gas is made up of molecules which areView Solution
- 4The relationship between pressure and the density of a gas expressed by Boyle’s law, $ P = KD$ holds trueView Solution
- 5View SolutionWe sit in the room with windows open. Then,
- 6This question has Statement $- 1$ and Statement $-2$. Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1$ : The internal energy of a perfect gas is entirely kinetic and depends only on absolute temperature of the gas and not on its pressure or volume.
Statement $2$ : A perfect gas is heated keeping pressure constant and later at constant volume. For the same amount of heat the temperature of the gas at constant pressure is lower than that at constant volume. - 7Under which of the following conditions is the law $PV = RT$ obeyed most closely by a real gasView Solution
- 8Consider two ideal diatomic gases $\mathrm{A}$ and $\mathrm{B}$ at some temperature $T$. Molecules of the gas $A$ are rigid, and have a mass $m$. Molecules of the gas $\mathrm{B}$ have an additional vibrational mode, and have a mass $\frac{\mathrm{m}}{4} .$ The ratio of the specific heats $(\mathrm{C}_{\mathrm{v}}^{\mathrm{A}}$ and $\mathrm{C}_{\mathrm{v}}^{\mathrm{B}})$ of gas $\mathrm{A}$ and $\mathrm{B}$, respectively isView Solution
- 9An ideal gas with adiabatic exponent $(\gamma=1.5)$ undergoes a process in which work done by the gas is same as increase in internal energy of the gas. The molar heat capacity of gas for the process is -View Solution
- 10Two moles of an ideal gas with $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}=\frac{5}{3}$ are mixed with $3$ moles of another ideal gas with $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}=\frac{4}{3} .$ The value of $\frac{\mathrm{C}_{\mathrm{P}}}{\mathrm{C}_{\mathrm{V}}}$ for the mixture isView Solution
