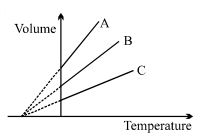
The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2 \,\,m$ at a pressure $2P$ is given by the straight line

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and its volume.View Solution
$Reason :$ The molecules of a gas collide with each other and the velocities of the molecules change due to the collision. - 2The molecules of a given mass of a gas have $r.m.s.$ velocity of $200 \,m s^{-1}$ at $27^o C$ and $1.0 \times 10^5 \,Nm^{-2}$ pressure. When the temperature and pressure of the gas are respectively, $127^o C$ and $0.05 \times 10^5 \,Nm^{-2},$ the $r.m.s.$ velocity of its molecules in $m s^{-1}$ isView Solution
- 3Two thermally insulated vessels $1$ and $2$ are filled with air at temperatures $({T_1},\,\,{T_2}),$ volume $({V_1},\,\,{V_2})$ and pressure $({P_1},\,\,{P_2})$ respectively. If the valve joining the two vessels is opened, the temperature inside the vessel at equilibrium will beView Solution
- 4View SolutionFor an ideal gas
- 5The temperature of $5$ moles of a gas which was held at constant volume was changed from ${100^o}C$ to ${120^o}C$. The change in internal energy was found to be $80$ Joules. The total heat capacity of the gas at constant volume will be equal to ...... $J/K$View Solution
- 6The equation of state for $5 \,g$ of oxygen at a pressure $P $ and temperature $T,$ when occupying a volume $V,$ will be (Where $R$ is the gas constant)View Solution
- 7For a diatomic gas change in internal energy for unit change in temperature for constant pressure and constant volume is ${U_1}$ and ${U_2}$ respectively. ${U_1}:{U_2}$ isView Solution
- 8The following sets of values for ${C_V}$ and ${C_P}$ of a gas has been reported by different students. The units are $cal/gm-mole-K.$ Which of these sets is most reliableView Solution
- 9At which temperature the velocity of $O_2$ molecules will be equal to the velocity of $N_2$ molecules at $0\,^oC$ ....... $^oC$View Solution
- 10${O_2}$ gas is filled in a vessel. If pressure is doubled, temperature becomes four times. How many times its density will becomeView Solution
