The following sets of values for ${C_V}$ and ${C_P}$ of a gas has been reported by different students. The units are $cal/gm-mole-K.$ Which of these sets is most reliable
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$5$ moles of oxygen is heated at constant volume from $10°C$ to $20°C.$ The change in the internal energy of the gas is (the gram molecular specific heat of oxygen at constant pressure...... $cal$ ${C_p} = 8$ $cal/mole \,°C$ and $R = 8.3 cal/mole\, °C)$View Solution
- 2View SolutionKinetic theory of gases was put forward by
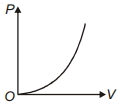
- 3The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
- 4The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 5One mole of monoatomic gas and three moles of diatomic gas are put together in a container. The molar specific heat (in $J\,{K^{ - 1}}\,mo{l^{ - 1}})$ at constant volume is $(R = 8.3\,J\,{K^{ - 1}}\,mo{l^{ - 1}})$View Solution
- 6View SolutionIf a Vander-Waal's gas expands freely, then final temperature is
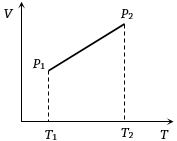
- 7From the following $V-T$ diagram we can concludeView Solution
- 8View SolutionThe specific heat relation for ideal gas is
- 9The temperature $(T)$ of one mole of an ideal gas varies with its volume $(V)$ as $T=-\alpha V^3+\beta V^2$, where $\alpha$ and $\beta$ are positive constants. The maximum pressure of gas during this process is ............View Solution
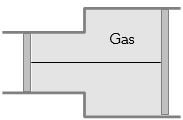
- 10View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will