The molecules of a given mass of a gas have $r.m.s.$ velocity of $200 \,m s^{-1}$ at $27^o C$ and $1.0 \times 10^5 \,Nm^{-2}$ pressure. When the temperature and pressure of the gas are respectively, $127^o C$ and $0.05 \times 10^5 \,Nm^{-2},$ the $r.m.s.$ velocity of its molecules in $m s^{-1}$ is
NEET 2016, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider the following statements for air molecules in an air tight container :View Solution
$(I)$ The average speed of molecules is larger than root mean square speed.
$(II)$ Mean free path of molecules is larger than the mean distance between molecules.
$(III)$ Mean free path of molecules increases with temperature.
$(IV)$ The rms speed of nitrogen is smaller than oxygen molecule.
Which of the above statements are correct?
- 2The ratio of total energy of all molecules of one mole $O_2$ to the total energy of all molecules of two moles of $He$ at the same temprature isView Solution
- 3View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
- 4A vessel contains $14\,g$ of nitrogen gas at a temperature of $27^{\circ}\,C$. The amount of heat to be transferred to the gap to double the r.m.s. speed of its molecules will be $......J$ $\left(\right.$ Take $R =8.32\,J\,mol ^{-1} k ^{-1}$ )View Solution
- 5View SolutionThe rate of diffusion is
- 6$1\ mole$ of a gas with $\gamma$ = $7/5$ is mixed with $1\ mole$ of a gas with $\gamma$ = $5/3$, then the value of $\gamma$ for the resulting mixture isView Solution
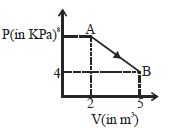
- 7A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
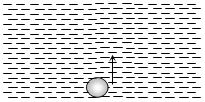
- 8An air bubble doubles its radius on raising from the bottom of water reservoir to be the surface of water in it. If the atmospheric pressure is equal to $10\, m$ of water, the height of water in the reservoir is ..... $m$View Solution
- 9A mixture of ideal gases has $2$ moles of He, $4$ moles of oxygen and $1$ mole of ozone at absolute temperature $T$. The internal energy of mixture is ............. $RT$View Solution
- 10A flask contains Hydrogen and Argon in the ratio $2: 1$ by mass. The temperature of the mixture is $30^{\circ} C$. The ratio of average kinetic energy per molecule of the two gases ( $K$ argon/ $K$ hydrogen) is: (Given: Atomic Weight of $Ar = 39.9$)View Solution
